The pharmaceutical industry continues to be a hotbed of innovation, with activity driven by the evolution of new treatment paradigms, gravity of unmet need, as well as the growing importance of technologies such as pharmacogenomics, digital therapeutics, and artificial intelligence. In the last three years alone, there have been over 633,000 patents filed and granted in the pharmaceutical industry, according to GlobalData’s report on Innovation in Pharmaceuticals: Coronavirus vaccine components. Buy the report here.
However, not all innovations are equal and nor do they follow a constant upward trend. Instead, their evolution takes the form of an S-shaped curve that reflects their typical lifecycle from early emergence to accelerating adoption, before finally stabilising and reaching maturity.
Identifying where a particular innovation is on this journey, especially those that are in the emerging and accelerating stages, is essential for understanding their current level of adoption and the likely future trajectory and impact they will have.
110 innovations will shape the pharmaceutical industry
According to GlobalData’s Technology Foresights, which plots the S-curve for the pharmaceutical industry using innovation intensity models built on over 756,000 patents, there are 110 innovation areas that will shape the future of the industry.
Within the emerging innovation stage, cell therapy for ocular disorders, coronavirus vaccine components, and DNA polymerase compositions are disruptive technologies that are in the early stages of application and should be tracked closely. Adeno-associated virus vectors, alcohol dehydrogenase compositions, and antibody serum stabilisers are some of the accelerating innovation areas, where adoption has been steadily increasing. Among maturing innovation areas are anti-influenza antibody compositions and anti-interleukin-1, which are now well established in the industry.
Innovation S-curve for the pharmaceutical industry
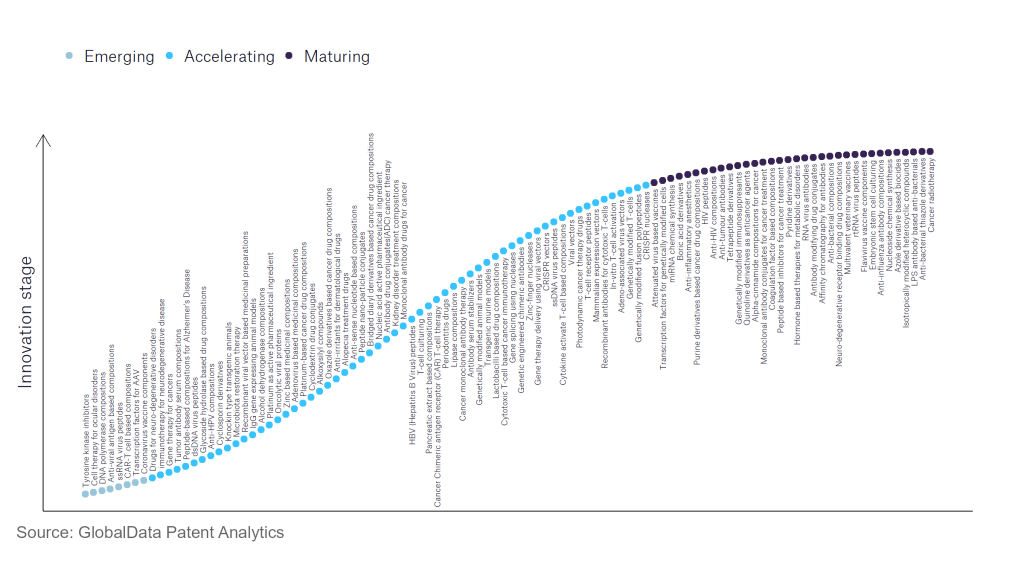
Coronavirus vaccine components is a key innovation area in pharmaceuticals
Components used in vaccines include antigens, preservatives, stabilisers, surfactants, residuals, diluents, and adjuvants. The key component in all vaccines is one or more active ingredients made from viruses or bacteria, also called antigens, which generates an immune response.
The active component of the Pfizer-BioNTech vaccine tozinameran, contains the genetic code for the coronavirus spike protein, inside a lipid (fat) capsule. The vaccine also contains other inactive ingredients such as cholesterol. The Oxford-AstraZeneca Vaxzevria vaccine is made from a modified adenovirus that delivers the genetic code for the coronavirus spike protein. The vaccine also contains inactive ingredients such as polysorbate 80, an emulsifier, a small amount of alcohol, and traces of magnesium (three to 20 parts per million).
GlobalData’s analysis also uncovers the companies at the forefront of each innovation area and assesses the potential reach and impact of their patenting activity across different applications and geographies. According to GlobalData, there are 50+ companies, spanning technology vendors, established pharmaceutical companies, and up-and-coming start-ups engaged in the development and application of coronavirus vaccine components.
Key players in coronavirus vaccine components – a disruptive innovation in the pharmaceutical industry
‘Application diversity’ measures the number of different applications identified for each relevant patent and broadly splits companies into either ‘niche’ or ‘diversified’ innovators.
‘Geographic reach’ refers to the number of different countries each relevant patent is registered in and reflects the breadth of geographic application intended, ranging from ‘global’ to ‘local’.
Patent volumes related to coronavirus vaccine components
| Company | Total patents (2021 - 2023) | Premium intelligence on the world's largest companies |
| Sanofi | 130 | Unlock Company Profile |
| C. H. Boehringer Sohn | 122 | Unlock Company Profile |
| Merck & Co | 76 | Unlock Company Profile |
| Takeda Pharmaceutical | 74 | Unlock Company Profile |
| Zoetis | 54 | Unlock Company Profile |
| US Department of Health and Human Services | 49 | Unlock Company Profile |
| Centre National de la Recherche Scientifique | 48 | Unlock Company Profile |
| Oswaldo Cruz Foundation | 45 | Unlock Company Profile |
| Novavax | 40 | Unlock Company Profile |
| Curevac | 40 | Unlock Company Profile |
| United Nations Industrial Development Organization | 37 | Unlock Company Profile |
| Regeneron Pharmaceuticals | 29 | Unlock Company Profile |
| Excivion | 22 | Unlock Company Profile |
| United States Of America | 21 | Unlock Company Profile |
| GSK | 20 | Unlock Company Profile |
| Moderna | 19 | Unlock Company Profile |
| Trustee for Horizon | 19 | Unlock Company Profile |
| U.S. Department of Defense | 19 | Unlock Company Profile |
| International Centre for Genetic Engineering and Biotechnology | 18 | Unlock Company Profile |
| Inovio Pharmaceuticals | 18 | Unlock Company Profile |
| Academia Sinica | 17 | Unlock Company Profile |
| Theravectys | 17 | Unlock Company Profile |
| Institut Pasteur | 17 | Unlock Company Profile |
| New York Blood Center | 15 | Unlock Company Profile |
| Reber Genetics | 15 | Unlock Company Profile |
| Johnson & Johnson | 14 | Unlock Company Profile |
| FDS Pharma LLP | 12 | Unlock Company Profile |
| Etubics | 12 | Unlock Company Profile |
| Erasmus MC | 11 | Unlock Company Profile |
| PerkinElmer | 11 | Unlock Company Profile |
| F. Hoffmann-La Roche | 11 | Unlock Company Profile |
| Ministry of Trade and Industry, Singapore | 10 | Unlock Company Profile |
| Trustee For Rope Pro Unit Trust | 10 | Unlock Company Profile |
| Guangzhou Anjie Biomedical Technology | 9 | Unlock Company Profile |
| CareGroup | 9 | Unlock Company Profile |
| TenGen Biomedical | 9 | Unlock Company Profile |
| Universidad El Bosque | 9 | Unlock Company Profile |
| US Government | 9 | Unlock Company Profile |
| Cellectis | 9 | Unlock Company Profile |
| NOF | 9 | Unlock Company Profile |
| Kitasato Daiichi Sankyo Vaccine | 8 | Unlock Company Profile |
| CanSino Biologics | 7 | Unlock Company Profile |
| iSD Immunotech | 7 | Unlock Company Profile |
| U.S. Department of Agriculture | 7 | Unlock Company Profile |
| Stichting Tech Wetenschapp | 7 | Unlock Company Profile |
| Sinovac Biotech | 6 | Unlock Company Profile |
| Valneva | 6 | Unlock Company Profile |
| American Type Culture Collection | 6 | Unlock Company Profile |
| Hawaii Biotech | 6 | Unlock Company Profile |
| IP Group | 6 | Unlock Company Profile |
Source: GlobalData Patent Analytics
Sanofi is the leading patent filer in coronavirus vaccine components. Sanofi is an innovative global healthcare company, whose primary focus is on rare diseases, rare blood disorders, neurology, immunology, and oncology. C. H. Boehringer Sohn and Merck are the other key patent filers in coronavirus vaccine components.
In terms of application diversity, Regeneron Pharmaceuticals is the top company, followed by Kitasato Daiichi Sankyo Vaccine and Excivion. By means of geographic reach, F. Hoffmann-La Roche held the top position, while TenGen Biomedical and Universidad El Bosque stood in the second and third positions, respectively.
To further understand the key themes and technologies disrupting the pharmaceutical industry, access GlobalData’s latest thematic research report on Pharmaceutical.
Data Insights
From

The gold standard of business intelligence.
Blending expert knowledge with cutting-edge technology, GlobalData’s unrivalled proprietary data will enable you to decode what’s happening in your market. You can make better informed decisions and gain a future-proof advantage over your competitors.







