The pharmaceutical industry continues to be a hotbed of innovation, with activity driven by the evolution of new treatment paradigms, and the gravity of unmet needs, as well as the growing importance of technologies such as pharmacogenomics, digital therapeutics, and artificial intelligence. In the last three years alone, there have been over 633,000 patents filed and granted in the pharmaceutical industry, according to GlobalData’s report on Innovation in Pharmaceuticals: Cyclosporin derivatives. Buy the report here.
However, not all innovations are equal and nor do they follow a constant upward trend. Instead, their evolution takes the form of an S-shaped curve that reflects their typical lifecycle from early emergence to accelerating adoption, before finally stabilising and reaching maturity.
Identifying where a particular innovation is on this journey, especially those that are in the emerging and accelerating stages, is essential for understanding their current level of adoption and the likely future trajectory and impact they will have.
110 innovations will shape the pharmaceutical industry
According to GlobalData’s Technology Foresights, which plots the S-curve for the pharmaceutical industry using innovation intensity models built on over 756,000 patents, there are 110 innovation areas that will shape the future of the industry.
Within the emerging innovation stage, cell therapy for ocular disorders, coronavirus vaccine components, and DNA polymerase compositions are disruptive technologies that are in the early stages of application and should be tracked closely. Adeno-associated virus vectors, alcohol dehydrogenase compositions, and antibody serum stabilisers are some of the accelerating innovation areas, where adoption has been steadily increasing. Among maturing innovation areas are anti-influenza antibody compositions and anti-interleukin-1, which are now well established in the industry.
Innovation S-curve for the pharmaceutical industry
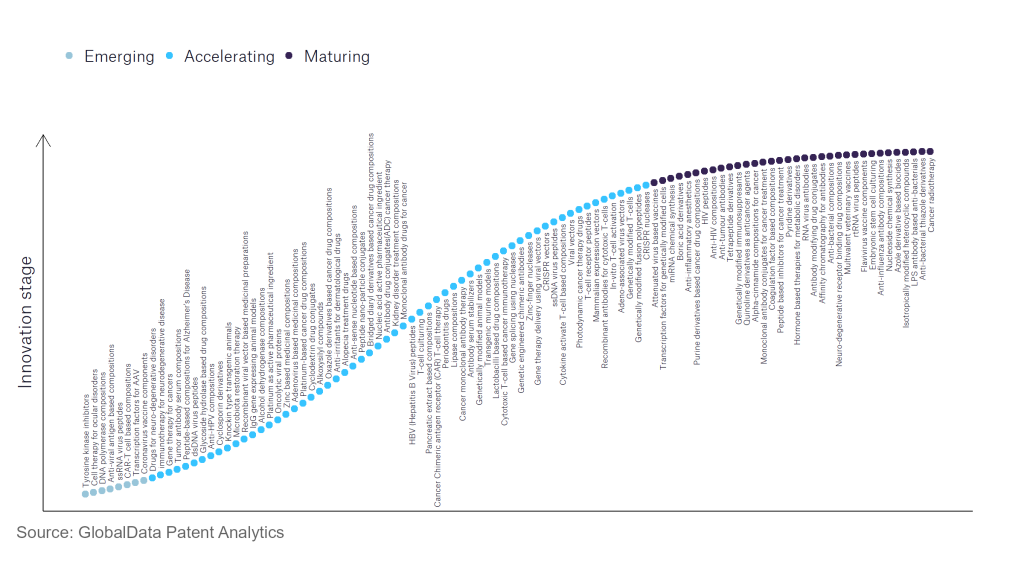
Cyclosporin derivatives is a notable innovation area in the pharmaceutical industry
Cyclosporin is a cyclic peptide composed of 11 amino acids and inhibits calcineurin. Cyclosporins are a family of neutral, highly lipophilic, and cyclic undecapeptides containing unusual amino acids. Cyclosporin is approved for a variety of indications. The drug is approved for the prophylaxis of organ rejection in allogeneic kidney, liver, and heart transplants. It is also used to prevent bone marrow transplant rejection. Cyclosporin can also treat patients with severe active rheumatoid arthritis (RA) who do not respond to methotrexate alone.
GlobalData’s analysis also uncovers the companies at the forefront of each innovation area and assesses the potential reach and impact of their patenting activity across different applications and geographies. According to GlobalData, there are 380+ companies, spanning technology vendors, established pharmaceutical companies, and up-and-coming start-ups engaged in the development and application of cyclosporin derivatives.
Key players in cyclosporin derivatives – a disruptive innovation in the pharmaceutical industry
‘Application diversity’ measures the number of different applications identified for each relevant patent and broadly splits companies into either ‘niche’ or ‘diversified’ innovators.
‘Geographic reach’ refers to the number of different countries each relevant patent is registered in and reflects the breadth of geographic application intended, ranging from ‘global’ to ‘local’.
Patent volumes related to cyclosporin derivatives
| Company | Total patents (2021 - 2023) | Premium intelligence on the world's largest companies |
| Immatics | 1297 | Unlock Company Profile |
| Philip Morris International | 406 | Unlock Company Profile |
| F. Hoffmann-La Roche | 358 | Unlock Company Profile |
| Bristol-Myers Squibb | 315 | Unlock Company Profile |
| Takeda Pharmaceutical | 234 | Unlock Company Profile |
| Protagonist Therapeutics | 228 | Unlock Company Profile |
| OncoTherapy Science | 225 | Unlock Company Profile |
| Amgen | 164 | Unlock Company Profile |
| Apellis Pharmaceuticals | 159 | Unlock Company Profile |
| Seagen | 152 | Unlock Company Profile |
| Bicycle Therapeutics | 150 | Unlock Company Profile |
| Centre National de la Recherche Scientifique | 148 | Unlock Company Profile |
| Merck & Co | 134 | Unlock Company Profile |
| Ironwood Pharmaceuticals | 125 | Unlock Company Profile |
| Ipsen | 118 | Unlock Company Profile |
| Caregen | 112 | Unlock Company Profile |
| Novo Nordisk Foundation | 111 | Unlock Company Profile |
| Ferring Holding | 109 | Unlock Company Profile |
| Aileron Therapeutics | 105 | Unlock Company Profile |
| GemVax & KAEL | 104 | Unlock Company Profile |
| UCB | 99 | Unlock Company Profile |
| Johnson & Johnson | 97 | Unlock Company Profile |
| Shanghai Techwell Biopharmaceutical | 94 | Unlock Company Profile |
| Forschungszentrum Julich | 92 | Unlock Company Profile |
| E. Merck | 92 | Unlock Company Profile |
| Sanofi | 90 | Unlock Company Profile |
| Palatin Technologies | 84 | Unlock Company Profile |
| Ministry of Trade and Industry, Singapore | 83 | Unlock Company Profile |
| Apitope International | 80 | Unlock Company Profile |
| Ambrx Biopharma | 75 | Unlock Company Profile |
| Vertex Pharmaceuticals | 74 | Unlock Company Profile |
| Equillium | 70 | Unlock Company Profile |
| MorphoSys | 70 | Unlock Company Profile |
| Novabiotics | 70 | Unlock Company Profile |
| Autoimmune Technologies | 69 | Unlock Company Profile |
| Pharma Mar | 68 | Unlock Company Profile |
| Administrators of Tulane Educational Fund | 62 | Unlock Company Profile |
| 3-D Matrix | 58 | Unlock Company Profile |
| AbbVie | 58 | Unlock Company Profile |
| Baxter International | 57 | Unlock Company Profile |
| Niox Group | 57 | Unlock Company Profile |
| United Nations Industrial Development Organization | 56 | Unlock Company Profile |
| Ecole Polytechnique Federale de Lausanne | 55 | Unlock Company Profile |
| AFFiRiS | 54 | Unlock Company Profile |
| Dr. Reddy's Laboratories | 52 | Unlock Company Profile |
| Zealand Pharma | 51 | Unlock Company Profile |
| Biocon | 51 | Unlock Company Profile |
| Xigen Inflammation | 50 | Unlock Company Profile |
| Cellivery Therapeutics | 47 | Unlock Company Profile |
| Pharis Biotec | 47 | Unlock Company Profile |
Source: GlobalData Patent Analytics
Immatics is the leading patent filer in cyclosporin derivatives. Immatics is a biopharmaceutical company focused on the development of T-cell immunotherapies to fight against cancer. Immatics is headquartered in Tuebingen, Germany. F. Hoffmann-La Roche and Bristol-Myers Squibb are the other key patent filers in cyclosporin derivatives.
In terms of application diversity, Western Sydney Local Health District (Australia) is the top company, followed by Warren Pharmaceuticals and Ipsen. By means of geographic reach, Ambrx Biopharma holds the top position. Adamed Pharma and BioMarin Pharmaceutical stand in second and third positions, respectively.
To further understand the key themes and technologies disrupting the pharmaceutical industry, access GlobalData’s latest thematic research report on Pharmaceutical.
Data Insights
From

The gold standard of business intelligence.
Blending expert knowledge with cutting-edge technology, GlobalData’s unrivalled proprietary data will enable you to decode what’s happening in your market. You can make better informed decisions and gain a future-proof advantage over your competitors.



