GlobalData Healthcare recently attended the World Evidence, Pricing and Access (EPA) congress in Amsterdam on 3-4 March 2026. This conference brings together key stakeholders from the pharma/biotech industry and payers to discuss the major topics that impact pharmaceutical pricing, reimbursement, and market access.
The congress opened with a plenary session that discussed and debated how to decode current disruptions seen in pharmaceutical markets globally. A major consensus was reached that instability is the new reality.
This is best reflected in an opening remark from Christoph Glaetzer, chief global value and access officer at J&J Innovative Medicine, that “it is time to unlearn what we have learned”.
During the congress, three key themes were explored:
MFN and its impact, notably on market access
The Most Favored Nation (MFN) policy was explained, analysed, and discussed in several sessions. The industry, along with policymakers, is continuing to gauge the impact the policy will have on market access and pharmaceutical expenditure, along with future investments in research & development.
Discussions were geared towards how to continue maximising revenues under an entirely new paradigm. Potential solutions emerged for ex-US pharmaceutical markets, including:
- Demonstrating greater value to HTA agencies to potentially secure higher prices
- Promoting the negotiations of prices and reimbursement attached to risk-sharing agreements
- Delaying or withdrawing products
- Launching in a more targeted indication.
Glaetzer added: “It’s no longer about the indication launch sequence. It’s about the entire portfolio of a company.” The comment demonstrates that pharmaceutical companies need to act differently and quickly.
This stance was further strengthened by a statement from Barry Farrimond, managing partner at ZS: “Winners don’t wait for instability to stop. They work fast in an unknown environment.”
However, the pharmaceutical/biotech industry is not the only stakeholder trying to navigate this series of unknowns. Patient advocacy groups have also voiced concern that the MFN policy will ultimately fall on patients, costing them chances to benefit from new and innovative treatments.
Artificial Intelligence (AI)
With an impressive 23 sessions dedicated to the subject, AI was undeniably one of the hottest topics of the congress.
The pharmaceutical and biotech industry highlighted that AI is heavily used nowadays in different activities, such as, but not limited to, clinical trials; commercial transformation; value dossier submissions; and HTA analysis.
Nevertheless, several speakers highlighted that the uptake was not as fast as expected and that manual efforts are still required.
Unsurprisingly, several key concerns were raised, including:
- Trust in data and accuracy (how to confirm hallucinations are not included in the model). On this topic, it was highlighted that human intervention is still required.
- Ethics: how to make sure terms and conditions from the sources are always met.
Joint Clinical Assessment (JCA)
The industry not only faces the hurdles of MFN policy and uptake of AI, but also new EU legislation in the form of JCAs. In January 2025, the JCA legislation came into force, requiring new oncology medicines as well as advanced therapies to be assessed under this new umbrella.
As seen in the graph below, 13 medicines were involved in JCA assessments in 2025.
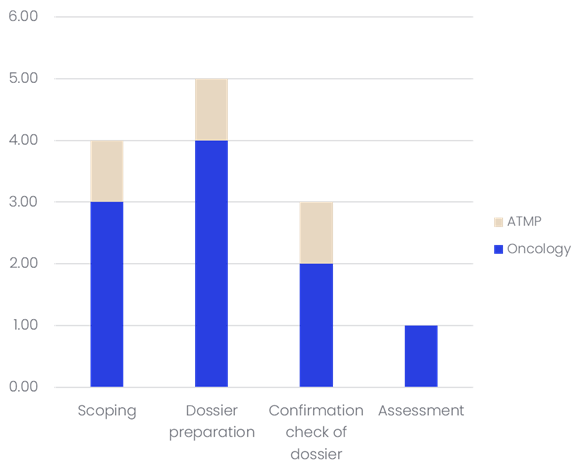
It is therefore no surprise that the JCA was a major focus of discussion, and that insights from its first year of implementation are in high demand.
As no dossier was fully finalised, many questions remain. Notably, population, intervention, comparison, and outcomes (PICO) submissions were reported to be challenging. Finding the right comparator is also not an easy task. Consequently, for each medicine assessed, many PICOs have to be submitted, thereby slowing the overall assessment.
The adoption of JCA recommendations by member states into national assessments was also discussed. Interestingly, most of the major EU member states reported that JCA assessments will be incorporated, while smaller markets have not yet decided if JCA will be used. This was quite a surprising outcome as initially, JCA outcomes were foreseen to be of high importance in smaller markets.
The 2026 EPA Congress was a success. Sessions and discussions generated great insights. GlobalData Healthcare will keep following up on these major topics. Stay tuned!
This article is produced as part of GlobalData’s Price Intelligence (POLI) service, the world’s leading resource for global pharmaceutical pricing, HTA and market access intelligence integrated with the broader epidemiology, disease, clinical trials and manufacturing expertise of GlobalData’s Pharmaceutical Intelligence Center. Our unparalleled team of in-house experts monitors P&R policy developments, outcomes and data analytics around the world every day to give our clients the edge by providing critical early warning signals and insights. For a demo or further information, please contact us here.





