Antimicrobial resistance (AMR) is a serious global health threat. A report commissioned by the UK Government and the Wellcome Trust – titled the Review on Antimicrobial Resistance and carried out by economist Jim O’Neill – concluded that each year 700,000 people die globally from drug-resistant bacterial and viral infections, and this figure looks set to rise to 10 million annually by 2050 if serious action is not taken.
Although AMR is not unique to antibiotics and bacteria, antibiotic resistance is of particularly serious and urgent concern as these drugs have become central to modern medicine, and resistance means the world is moving back to a situation where common infections could become untreatable.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The World Health Organization’s (WHO) World Antibiotics Awareness Week, which runs between 18 and 24 November this year, continues to remind the world about antibiotic resistance and why it is a threat to global health.
It notes what individuals, as well as health workers and policy makers, can do to prevent the further spread of antibiotic- resistant bacteria, but there is also a need to develop new drugs that can target bacteria resistant to existing antibiotics. However, incentivising pharmaceutical companies to invest in ongoing research and development (R&D) efforts has been a huge challenge to governments and international organisations.
Challenge of multi drug resistance
Even more threatening and challenging than bacteria being resistant to one antibiotic are multi-drug resistant bacteria. Examples are gram-negative bacteria that produce the enzyme extended spectrum beta lactamase, such as E. Coli and K. pneumoniae.
These types of bacteria have been listed by the WHO as priority pathogens,; moving the AMR focus beyond just gram-positive bacteria, like methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant Enterococci, and mycobacterium tuberculosis.
Multi-drug resistance occurs either because a single bacterium has several resistant genes, which accumulate on a plasmid, or there is a single resistance mechanism linked with multiple antibiotics.
Multi-drug resistant bacteria significantly contribute to the spread of AMR because when a multi-drug resistant plasmid is transferred between two bacteria it passes on resistance to multiple drugs.
This means the pool of effective treatments is being reduced even further and makes the need for new drugs to overcome resistance even more urgent.
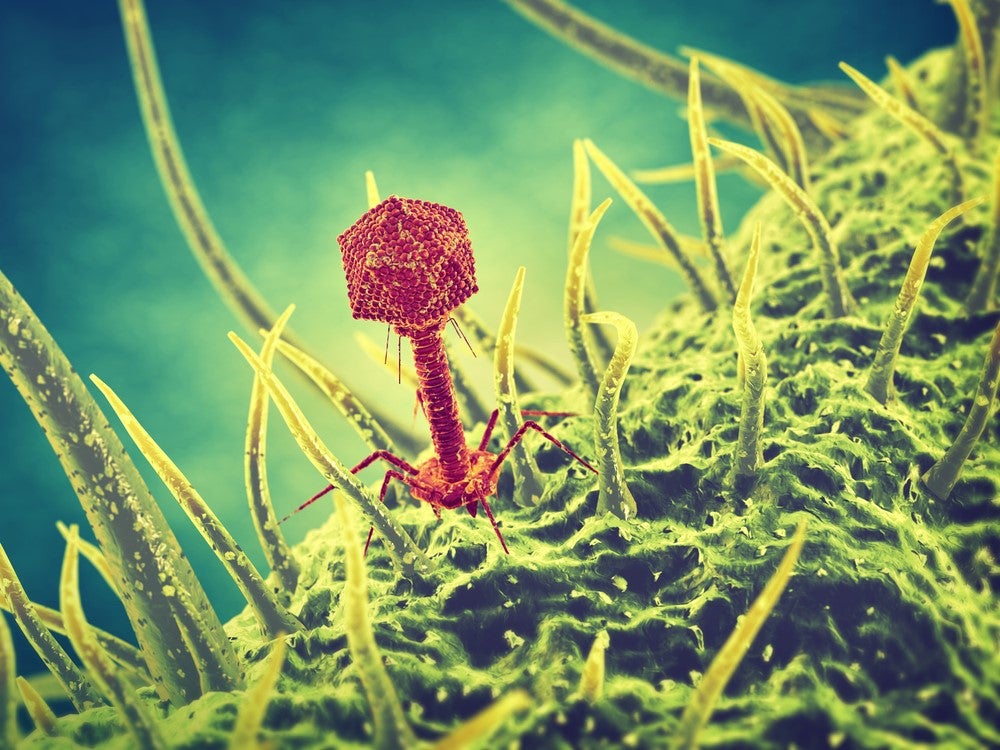
Beyond antibiotics: promise of bacteriophages
One of the main priorities in developing therapeutic agents against multi-drug resistant bacteria must be to ensure the bacteria do not simply become resistant to the new products, taking researchers back to square one. Instead, new treatment paradigms need to be created against bacteria.
Bacteriophages represent an interesting source of new antibacterial approaches. Used in various infectious diseases since the early 1900s, manufacturing and purification processes for phage-based therapies have been refined over time, and now regulators, including the FDA and EMA, are beginning to view phage-based therapies as a possible alternative where bacteria have developed resistance to antibiotics.
Phages – viruses that kill specific bacteria – have many advantages over traditional antibacterial drug approaches: they co-evolve with bacteria, creating rich sources of phages that can work against the bacteria and they do not produce toxic effects as they regulate themselves at the site of infection, according to a 2018 paper in Surgical Infections.
Using phages in the clinic
One example of phages being used in clinical settings to treat multi-drug resistant bacterial infections was a case in 2018, when doctors at the Howard Hughes Medical Institute (HHMI) in Chevy Chase, Maryland created a personalised, genetically engineered bacteriophage treatment to treat a 15-year-old patient’s bacterial infection complications from a lung transplant for her cystic fibrosis.
Using a collection of phages from global locations at HHMI for biologic study, they tested individual phages known to infect bacterial relatives of the patient’s strains and then genetically altered them so they were more aggressive against the bacterial cell. Then a mix of these genetically modified phages were administered through an intravenous infusion to the patient simultaneously.
Published in Nature Medicine, the patient’s results showed that over six months, skin nodules caused by bacterial infections disappeared, her surgical wound closed up and her liver function improved. Most importantly, over a year later, the bacteria have not shown any signs of developing resistance to the phages administered.
HHMI professor at the University of Pittsburgh Graham Hatfull explained: “Such a treatment could offer a personalized approach to countering drug-resistant bacteria. It could even potentially be used more broadly for controlling diseases like tuberculosis.
“We’re sort of in uncharted territory. But the basics of the young woman’s case are pretty simple: we purified the phages, we gave them to the patient, and the patient got better.”




