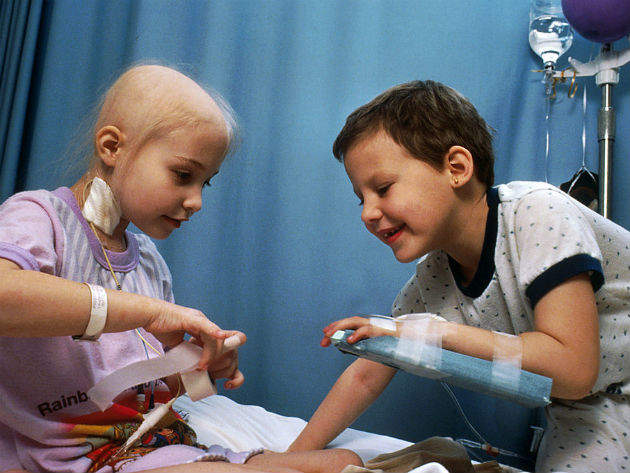

Cancer experts are calling on European officials to reform outdated rules on clinical trials that allow pharmaceutical companies to opt out of testing new cancer drugs in children even where there is good evidence they could be beneficial.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Although the aim of the EU Paediatric Regulation, which was introduced in 2007, was to improve children’s access to new treatments, a loophole means that companies can apply for a waiver from having to trial a cancer drug in children if it targets a cancer that does not affect them, such as lung cancer – even if the drug’s mechanism of action means it could be effective.
And apply they most certainly have. Indeed, a new analysis by The Institute of Cancer Research (ICR) of EMA data shows that over the last five years (2012-2016) pharmaceutical companies were granted waivers from having to trial cancer drugs in children for 62% of cancer treatments that were ultimately approved. Many of these, such as axitinib and obinutuzumab, have a mechanism of action that is potentially relevant to children’s cancers.
Crucially, the ICR is also insisting that if the rules were to be reformed, these changes would need to be applied across Europe so children in the UK do not lose out on access to the latest treatments after leaving the EU.
Pharma Technology Focus sits down with the institute’s senior policy and engagement manager Dr Eva Sharpe to find out why the time for reform is now and how European cancer trial regulations need to change to better serve young patients.
Elly Earls: The ICR is campaigning for reforms of the regulation of European cancer trials for children. Can you explain why this is such as important issue for the institute?
Dr Eva Sharpe: Dr Eva Sharpe: The ICR has been campaigning for several years to change European regulations so more cancer drugs are evaluated in children. We are seeking reform to the Paediatric Regulation – a piece of legislation that governs clinical trials in children across Europe – to make sure drug companies always test new drugs in children whenever there is evidence that they could be beneficial. At the moment the regulation contains a loophole which means companies that have developed drugs for adult cancers can often avoid running trials in children. If we close this loophole, there’s the potential to make a huge difference for children with cancer by giving them access to many more of the latest treatments in clinical trials.
EE: How would you like to see the regulations updated and why?
ES: We want to see the Paediatric Regulation updated to take into account the latest in cancer science, which has shown us that many cancer drugs for adults can be beneficial for children’s cancers too.
At the moment, the Paediatric Regulation is meant to require companies to plan how they’ll study a new drug in children before they can gain marketing authorisation in adults. However, companies can get a waiver from having to do this if for example a drug has been developed for an adult disease that doesn’t occur in children.
But for cancer treatments this system isn’t really working, as modern cancer drugs rather than being designed to combat a particular tumour type, such as breast cancer or lung cancer, are often instead targeted against specific molecular defects. These defects are normally present in just a subset of each tumour type, but they may exist across several cancer types, including both adults’ and children’s cancers.
A drug may be relevant for several different types of cancers – in both adults and children – if the specific molecular defect driving the cancer is the same. Under the current system, companies are being granted waivers from having to test adult cancer drugs in children, even though their mechanism of action means they could be beneficial.
We want to see the regulation updated so that cancer drugs are tested in children whenever their mechanism of action suggests they could be effective for children’s cancers. This single change would make a massive difference to the numbers of trials opening for children and could speed up access to innovative new treatments.
We were pleased to see the EMA make changes to its guidance on the Paediatric Regulation a couple of years ago that may reduce the number of waivers granted when it comes into force next year. But this is only an interim solution. Until the Regulation itself is updated, we’re not going to see the size of increase in trials for children that we need.
EE: Why is it so important to address this issue now?
ES: We currently have our best chance yet for the Regulation to be updated. It’s now 10 years since it came in to force and the European Commission is preparing a report on its impact. It is a major opportunity for the Commission to look at the regulation in detail and listen to evidence, from organisations like the ICR, that the current system is not working. We’re really hoping the Commission will agree that an update is needed and take this opportunity for change. Under the current outdated Regulation, children across Europe are missing out on the advances that adults are seeing in cancer treatment.
EE: How important for British children is it that this reform is applied across Europe, and why?
ES: As childhood cancers are rare, we can’t run trials in just one country – to include sufficient numbers of patients, trials need to bring together multiple different centres internationally. Regulations therefore need to be consistent across borders so that the different collaborating countries can all work together under the same rules. When the UK leave the EU, sustaining these international collaborations will be vital to ensure British children can continue to access Europe-wide trials. That means that it is no use changes to regulation being applied only in the UK – they have to be implemented across Europe.
EE: If the law were to be reformed, how much could it improve survival rates in childhood cancers?
ES: At the moment, very few cancer drugs are developed specifically for children, and with the current regulation, the advances that we are seeing in adult cancer treatments aren’t happening for children anywhere near fast enough. To make a real difference in survival rates for children with cancer we need a major expansion in the number of cancer medicines available for children, and this can only happen by running more trials.




