Kisspeptin, a hormone produced in the hypothalamus, holds diverse clinical potential that researchers are only now starting to understand. The story starts with the 2003 discovery that the hormone is vital for both humans and animals to go through normal puberty, which led to the realisation that kisspeptin stimulates reproductive hormones.
As well as its identification as a potential suppressor of cancer metastasis (hence its original name, metastin), researchers are getting to grips with kisspeptin’s important role in the human reproductive system and the attendant opportunities for new therapies targeting sexual and reproductive disorders.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Professor of endocrinology and metabolism Waljit Dhillo and his team at Imperial College London have been investigating the clinical applications of kisspeptin for more than a decade, having demonstrated kisspeptin’s reproductive hormone-stimulating effect in humans for the first time in 2005.
Dhillo and his colleagues have trialled kisspeptin as an alternative to human chorionic gonadotropin (hCG) in women undergoing in-vitro fertilisation (IVF) who are at a heightened risk of ovarian hyperstimulation syndrome (OHSS), with a successful clinical study published in 2015.
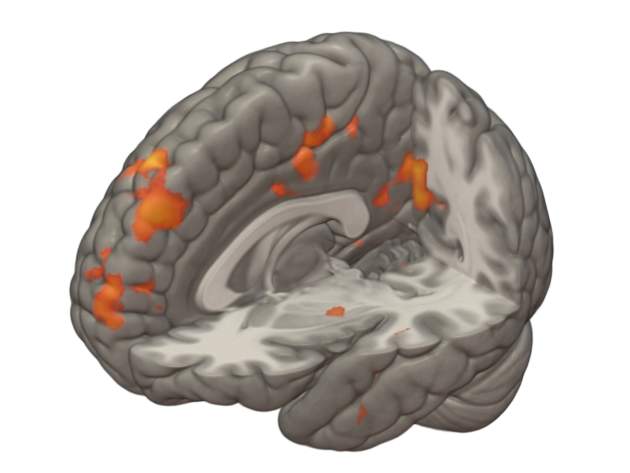
More recently, the team has also demonstrated – during a trial on a group of 29 healthy male volunteers – that kisspeptin can enhance neural processing of sexual, romantic and emotional images, potentially opening up the hormone’s applicability to psychosexual disorders and associated fertility problems among couples, and even as a new treatment for depression.
We spoke to Dhillo to discuss the clinical potential of the ‘love hormone’, how research is progressing, and the roadmap for translating these discoveries from the laboratory to the clinic.
Chris Lo: Could you give me an update on how your research into kisspeptin for IVF has progressed in the last year or so, and where it is at currently?
Waljit Dhillo: hCG is the commonly-used trigger in IVF and it works and it’s very good for maturation, but it acts for too long, it’s non-physiological and hence can give you what’s called ovarian hyperstimulation syndrome [OHSS], and that can cause a number of deaths, plus illness in women each year having IVF. We know what the cause is – it’s the drug that’s used in IVF.
hCG acts directly on the ovaries, so one of the advantages you’ve got with kisspeptin is that it’s acting up in the hypothalamus on what’s called GnRH [gonadotropin-releasing hormone], and that then acts on the pituitary, which then acts on the gonad. So it’s a more indirect and a more physiological way to stimulate [ovulation].
So the idea, going back a few years, was does it work? Yes it does. And then the next thing was does it work in high-risk women? And the papers published in 2015 showed that with the highest-risk women, we were getting very little OHSS, and were getting good efficacy. We’ve just moved to another study that we’ve just finished, which will hopefully come out soon, looking at tweaking whether a single or a double dose of kisspeptin is better, and a double dose looks like it will be better. So we’re now moving forward to take that through to the clinic.
CL: What implications could your findings on kisspeptin’s effect on sexual and emotional brain processing have from the perspective of human reproductive, sexual, and mental health?
WD: In the last ten years people have been looking at the reproductive pathway and the reproductive hormones, but we’re all aware that the brain is involved in how we perceive sexual stimuli. It was known for a while that kisspeptin receptors are also found in lots of other areas of the brain, not just where the reproductive hormones are. They’re found in the limbic system, which is involved in reward and how the brain perceives sexual images.
The obvious thing then was to see whether kisspeptin was affecting these areas. The first thing we did a few years ago was look in animals – when we gave them kisspeptin, their amygdala activity changed, which is one of the areas of the limbic system. Following that, the next step was to look in humans, and we found changes again when they had kisspeptin versus saline when they were looking at sexual and bonding pictures. It was much higher than when they looked with saline, and it correlated with their behavioural score. So this was the first demonstration of a link between the hormone and sexual and emotional brain processing.
CL: What work still needs to be done to confirm and validate these findings?
WD: The implications for patients, and we will need to do those studies, would be if you’ve got a problem with psychosexual disorders and how you’re processing sexual images, then this can obviously be very useful. That affects up to one in three people at various stages, and if you could give them a therapy that would improve that, that would obviously help their relationships and in terms of infertility patients, it could help a high proportion of those as well. We’ve only done the study on healthy males; we now need to move to females and then to patients with those disorders as we move forward.
CL: How do you speculate that kisspeptin, if used as a treatment for psychosexual disorders or couples having trouble conceiving, would break from the norm in terms of the current standard of care in this field?
WD: Well, the current standard of care is things like Viagra, which is basically a vasodilator and a mechanical [treatment], [stimulating] more blood flow down to the genital area. So it’s not addressing the problem, it’s kind of using an alternative mechanism to try and help. It actually often doesn’t work, because if you’ve got psychological issues in terms of how you’re perceiving things, then more blood flow to the genital area is not going to particularly help when the problem’s somewhere else. This may address or improve, if the studies continue to be positive, the underlying deficit, because that’s where the problem seems to be.
CL: Given the apparent effect of kisspeptin on regulating negative mood, is there any indication that it could be a worthwhile alternative to traditional antidepressants such as selective serotonin reuptake inhibitors (SSRI)?
WD:
There’s a paper on depression in an animal model where kisspeptin administration seems to improve their depression scores as well. So that is an area that, again subject to funding, we will be looking forward to as it’s obviously a big problem.
It’s too early to tell really, because we’d need to see what the efficacy was. When we saw that change in negative mood, we then looked at the literature and honestly there was that paper on depression. So the next stage would be a specific study to look at negative scores, and then in patients with depression to see whether their symptoms improved. Obviously, that takes time.
CL: Where do you think we are in the journey of discovering the clinical potential of kisspeptin?
WD: I think time will tell, and the proof’s in the pudding. Until you actually do the studies, you don’t know. So far with the IVF work it’s gone very well. Five or six years ago we were hypothesising about whether it works, now we’re tweaking it to make it as good as we can get before we do a head-to-head study.
The study we’ve just published in terms of sexual and emotional processing, it’s the first time it’s ever been shown that this hormone can link the two. But it’s taken about 18 months to get there doing the studies, because you have to do the studies rigorously and robustly to get a reliable answer. But now that we’ve got those, the next stage is to take each part forward.
CL: Have any red flags been raised during the studies about potential adverse effects of kisspeptin administration?
WD: That’s been one of the encouraging things. The doses we’re using are incredibly low, because it’s essentially not a drug, it’s a hormone that all of us have had to get us through puberty, it’s a hormone that, in normal pregnant women, their placenta churns out thousands-fold higher [than the dose], and the baby that’s developing sits in thousands of picomoles per litre of kisspeptin. And we’re using very small doses to get these effects.
There is some animal data and in-vitro data showing that if you give very, very high doses you can get some side effects, but, touch wood, at the doses we’ve given, and we’ve probably administered it to several hundred people now, it’s pretty clean. So it’s been really encouraging, and that’s one of the reasons why we’ve kept going.
CL: Have you considered a spin-out company to translate or commercialise treatments derived from the hormone?
WD:
Yeah, that’s a possibility. It’s not something that we’ve done yet because we’re proving efficacy. What we’re trying to do is [work out] all the different indications. If you’re looking for a shorter profile are you going to need a different agent compared to if you’re looking for a longer profile? So for the IVF we definitely don’t want a very long preparation because that’s the problem with hCG. If we’re going for depression we might want a longer preparation that lasts 12 or 24 hours so you only have to give it once a day.
So that’s where we’re heading, first using the endogenous molecule to work out the physiology. Once you know what you’re then looking for, you can then spin out and try to develop analogues that suit that indication. I think the amazing thing for us is the specificity and the lack of side effects. The doses you need are very small to have a big effect. That’s been the biggest thing, I think. Certainly within the next five years we’re aiming to be, in the most interesting areas, heading towards the clinic with full-scale clinical trials.





