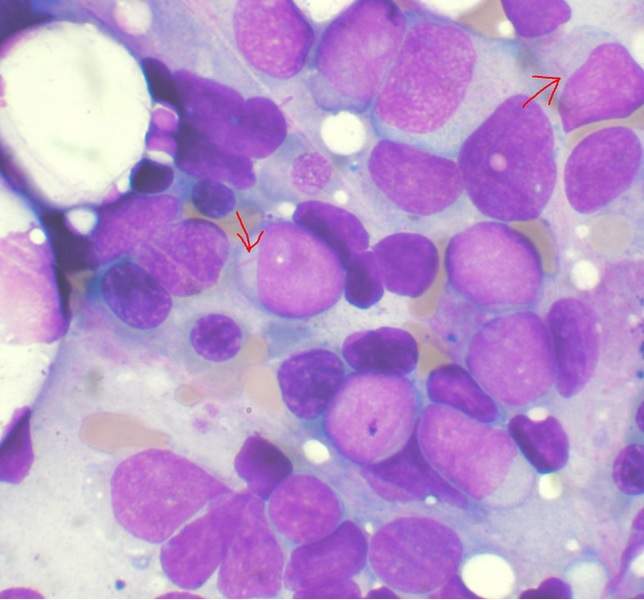
The US Food and Drug Administration (FDA) has approved Pfizer’s Daurismo (glasdegib) indication to treat patients with newly diagnosed acute myeloid leukaemia (AML).
The once-daily oral medicine is the first and only FDA-approved Hedgehog pathway inhibitor for AML.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
It is taken in combination with low-dose cytarabine (LDAC), a form of chemotherapy and can be used to treat AML in patients aged 75 years or older.
AML is a rapidly progressing bone marrow cancer with poor survival rates when compared to other leukaemias.
Pfizer Oncology global president Andy Schmeltz said: “As our second medicine approved in the last 14 months for patients with acute myeloid leukaemia, Daurismo reinforces our commitment to delivering new medicines to patients living with some of the most difficult-to-treat cancers, especially those for which there are limited treatment options available.
“We are proud to now offer these patients for whom intensive chemotherapy is not an option a new oral medicine, taken in combination with low-dose chemotherapy, that may improve their chances of survival.”
The FDA’s approval is based on a pivotal, randomised, international Phase II BRIGHT 1003 trial, which enrolled 115 patients with newly diagnosed AML.
Patients were randomised 2:1 to receive Daurismo plus LDAC or LDAC alone.
Pfizer Global Product Development oncology chief development officer Mace Rothenberg said: “Daurismo, a Hedgehog pathway inhibitor, was discovered in Pfizer laboratories and exemplifies our continued commitment to developing medicines that have the potential to advance cancer therapeutics.
“The ongoing Phase III BRIGHT trials are evaluating Daurismo in combination with other agents commonly used to treat patients with acute myeloid leukaemia, in an effort to understand the full potential of this medicine against this aggressive leukaemia.”




