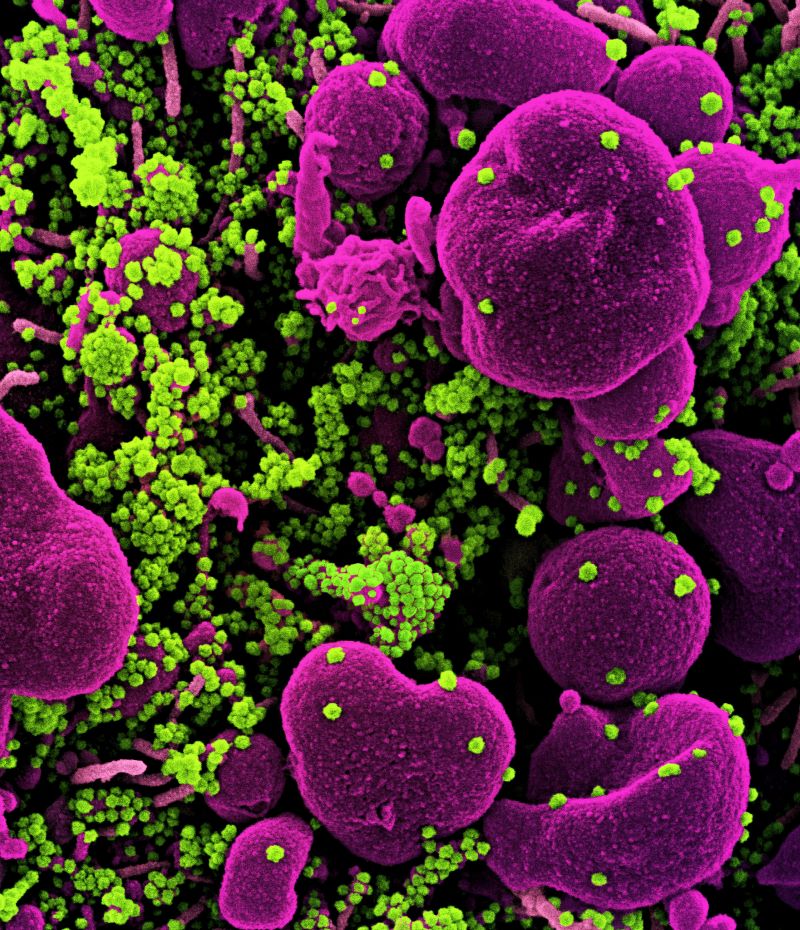
Kinarus Therapeutics has reported that its KIN001 offered robust antiviral efficacy against the original strain and variants of concern (VOC) of the SARS-CoV-2 virus in vitro.
The partnership between virologists at Kinarus and the Friedrich-Alexander-Universität Erlangen-Nürnberg (FAU) demonstrated that KIN001 could fully hinder replication of the virus in various human cell lines for all SARS-CoV-2 virus strains, including VOC such as Delta and Omicron.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The company had obtained an up to $7.2m (CHF7m) grant from the Programme for COVID-19 medicines of the Swiss Federal Office of Public Health to back the clinical development of KIN001.
A Phase II KINETIC clinical trial of KIN001 for the treatment of hospitalised Covid-19 patients had enrolled 130 subjects by 6 June this year.
Interim data from the trial are anticipated to be available in the third quarter of this year.
Another Phase II KINFAST trial of KIN001 for treating ambulatory Covid-19 patients had recently obtained regulatory authorisation in Switzerland and Germany and will begin soon.
The effects of KIN001 are expected to fight the virus in three complementary ways: anti-viral, anti-fibrotic, and anti-inflammatory.
Anti-viral could avert viral replication, anti-inflammatory effects could lower the inappropriate inflammatory response of the body and anti-fibrotic effects could boost the healing of damaged tissues, thereby expediting recovery and lowering the chances of long Covid symptoms.
As against vaccines, antibody and nucleic acid-based treatments that act on viral proteins, KIN001 acts on the host cell pathways required for the replication of the virus.
The VOCs that may arise in future are less likely to avoid this mechanistic difference.
Kinarus Therapeutics CEO Alexander Bausch said: “It is highly encouraging that equivalent potency of KIN001 was observed against SARS-CoV-2 and the VOCs, including delta and omicron.
“This leads to the conclusion that KIN001 will also likely demonstrate anti-viral properties against future variants.
“This bodes well for the ongoing Phase II KINETIC study of KIN001 in hospitalised Covid-19 patients; we look forward to the interim data around the end of the third quarter.”




