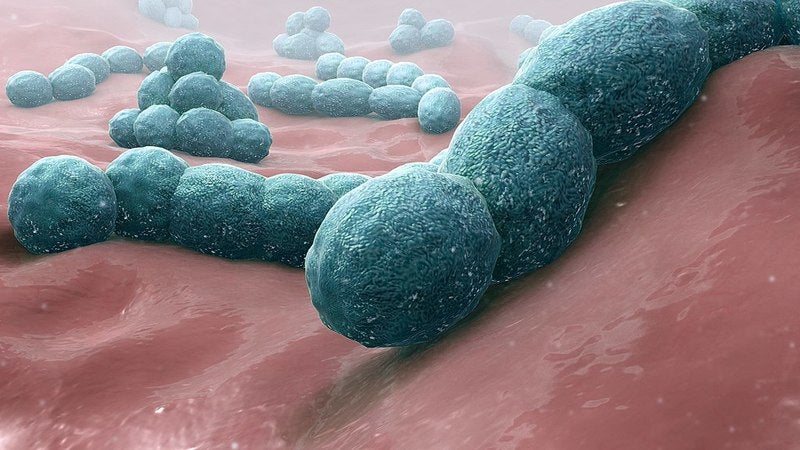
Merck has secured breakthrough designation from the US Food and Drug Administration (FDA) for its investigational 15-valent pneumococcal conjugate vaccine, V114.
The company is developing the vaccine to prevent invasive pneumococcal disease (IPD). The FDA granted the designation for its use in paediatric patients aged six weeks to 18 years.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
V114 is also under development for the prevention of IPD in adults; the vaccine is currently being assessed in Phase III clinical trials.
The FDA granted the drug breakthrough status after reviewing immunogenicity data from Study 005 and 008.
A Phase I/II, multi-centre, randomised, double-blind trial, Study 005 investigated the safety, tolerability and immunogenicity profiles of four different lots of a new V114 formulation in healthy adults and infants.
Study 008 was a Phase II, proof-of-concept trial performed to confirm the Study 005 findings in a larger population of infants.
In both the trials, the vaccine was found to trigger an immune response in infants for 22F and 33F disease-causing serotypes that are not contained in the existing 13-valent pneumococcal conjugate vaccine.
V114 also showed non-inferiority for the serotypes present in both vaccines.
Merck Research Laboratories vaccine and infectious diseases clinical research senior vice-president and head Nicholas Kartsonis said: “We are pleased with the data on V114 compiled to date, and we look forward to working closely with the FDA on the subsequent development of this investigational vaccine.
“The goal of our programme in paediatric patients remains focused on providing additional serotype coverage versus currently available vaccines, while at the same time maintaining a strong immune response across all serotypes in the vaccine.”
Caused by Streptococcus pneumonia, IPD results in a variety of clinical syndromes such as bacteremia and meningitis. The disease is known to lead to notable morbidity and mortality in the US.
According to the US Centers for Disease Control and Prevention (CDC), launch of 7-valent pneumococcal conjugate vaccine in 2000 decreased the rates by 64-77% in adults and older children, and dropped to less than one case per 100,000 among children under five years of age.
The rates were observed to lower further in 2010 with the availability of 13-valent pneumococcal conjugate vaccine. However, more than 35,000 cases and 4,200 deaths were still recorded in 2011.




