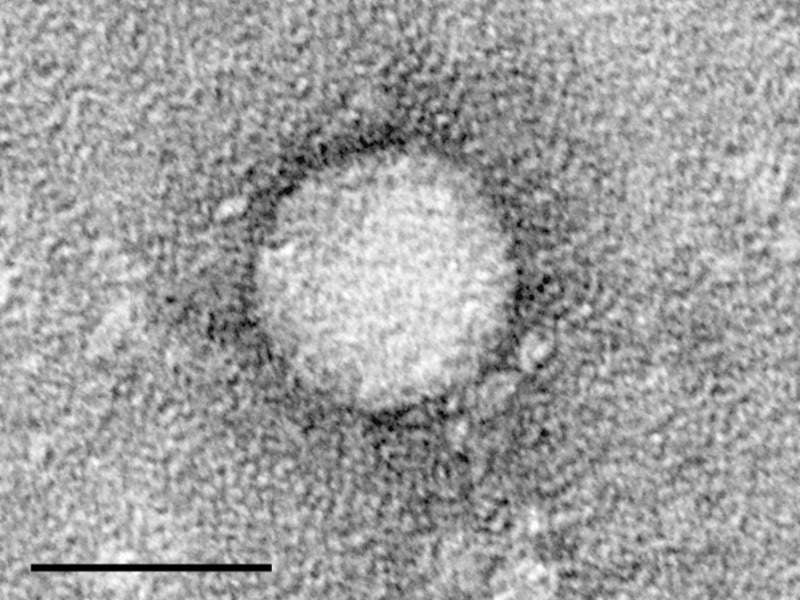

Public health organisation Medicines Patent Pool (MPP) has entered a new licence and technology transfer agreement with Egyptian company Pharco Pharmaceuticals for ravidasvir (RAV).

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
RAV is an investigational direct-acting antiviral (DAA) that can be administered in all six major hepatitis C genotypes.
Medicines Patent Pool executive director Greg Perry said: “If successfully developed, ravidasvir could help treat millions of hepatitis C patients and we are pleased to work with Pharco to ensure the availability of affordable versions.”
The agreement will enable competitive supply of RAV in low and middle-income countries (LMICs), including high-prevalence nations such as Russia, Ukraine, Egypt and Iran.
Pharco Pharmaceuticals CEO Dr Sherine Helmy said: “Chronic hepatitis C affects approximately 71 million people globally, with Egypt suffering from one of the highest burdens.
“RAV, in combination with other hepatitis C treatments, could support new national as well as global goals to eliminate the virus.”
This agreement expands the geographic scope of a licence to several new LMICs.
The previous agreement signed in March last year by Presidio, the original developer of RAV and the Drugs for Neglected Diseases initiative (DNDi), had limited geographical scope of the licence.
The combined MPP and DNDi agreements are expected to benefit 85.3% of the total hepatitis C patients who live in the 139 block of economies classified by the World Bank as low-income and middle-income.
DNDi executive director Dr Bernard Pécoul said: “DNDi is currently sponsoring trials of ravidasvir in combination with sofosbuvir with the objective of providing a pan-genotypic and affordable treatment for eventual distribution in developing countries.
“DNDi welcomes the opportunity to work with all partners to ensure that safe, effective and affordable drugs are made available to all those in need.”
Pharco previously conducted a Phase III trial of RAV in Egypt that demonstrated cure rates of up to 100% in patients with genotype 4 when RAV was used in combination with other treatments.
The MPP-Pharco licence for RAV will provide royalty over net sales in the licensed territory (4% in low-income nations and 7% for middle-income countries).
The licence includes a technology transfer to MPP's sub-licensees.
Image: Electron micrographs of hepatitis C virus purified from cell culture. Photo: courtesy of the Center for the Study of Hepatitis C, The Rockefeller University / Maria Teresa Catanese, Martina Kopp, Kunihiro Uryu and Charles Rice.




