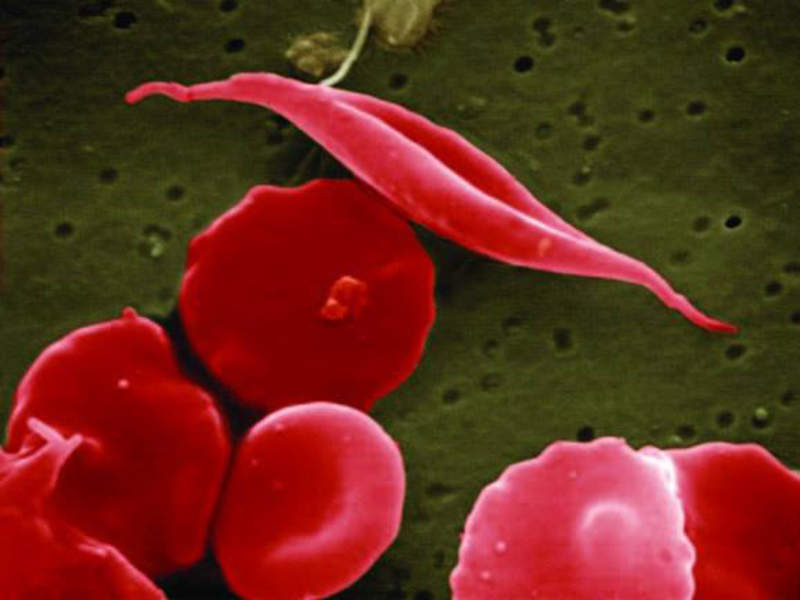
The National Institutes of Health (NIH) has launched a new initiative to expedite the development of genetic therapies to treat sickle cell disease.
The Cure Sickle Cell Initiative will leverage new genetic discoveries and technological advances to safely progress the ‘most promising’ therapies into clinical trials within five to ten years.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
A genetic blood disorder, sickle cell disease is characterised by clogged blood vessels and oxygen deprivation that can lead to organ damage, severe pain and premature death.
Over the years, basic research has resulted in new curative genetic approaches such as genetic editing of bone marrow cells.
Under the Production Assistance for Cellular Therapies (PACT) programme, NIH’s National Heart, Lung, and Blood Institute (NHLBI) is working on producing cellular therapeutic products, including genetically modified cells.
With the new initiative, NHLBI will support the development of cell and genetic therapies resources, clinical trials, analyses of various management approaches, data repositories, and patient and advocate engagement activities.
Every year, NIH spends around $100m on sickle cell disease research. This year, NHLBI plans to offer an additional $7m to commence the initiative’s research and engagement infrastructure.
NHLBI director Gary Gibbons said: “Our scientific investments have brought us to a point where we have many tools available to correct or compensate for the defective gene that causes sickle cell disease. We are now ready to use these tools to speed up our quest for a cure.”
The initiative and its research partners intend to build a data warehouse of genetic therapies for the disease. They will also perform comparative analyses of therapeutic strategies in order to investigate clinical and cost effectiveness.
In addition, they plan to establish national networks to provide patients and providers with easy access to the research, clinical trials and other activities across the country.




