A study funded by the National Institutes of Health’s (NIH) National Eye Institute (NEI) has successfully reversed congenital blindness by changing the supportive Müller glia cells in the retina into rod photoreceptors.
Conducted in mice, the research is expected to help in advancing regenerative therapies for the treatment of blinding diseases, such as retinitis pigmentosa.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Photoreceptors are light-sensitive cells in the retina that signal the brain upon activation. In mammals, including humans, photoreceptors cannot regenerate on their own.
NEI retinal neuroscience programme director Thomas Greenwell said: “Rods allow us to see in low light, but they may also help preserve cone photoreceptors, which are important for colour vision and high visual acuity. Cones tend to die in later-stage eye diseases.
“If rods can be regenerated from inside the eye, this might be a strategy for treating diseases of the eye that affect photoreceptors.”
During the initial phase of the study, the researchers injected a gene for activating the beta-catenin protein in order to trigger Müller glia in normal mice.
After certain duration, these mice were injected in the eye with factors required to stimulate development of the newly divided cells into rod photoreceptors.
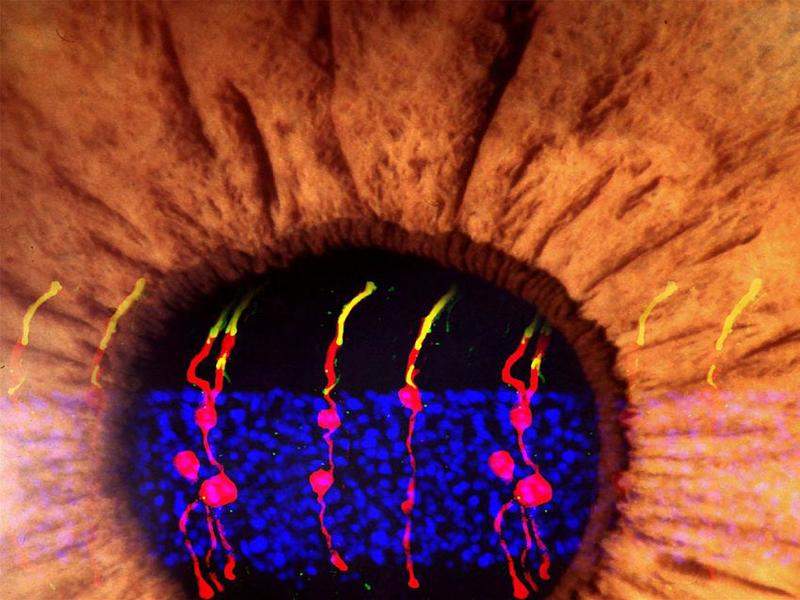
Microscopy was used to visually track the newly formed cells. It was observed that the new rod photoreceptors were similar in structure to the original photoreceptors.
The new cells were also able to form synaptic structures that enable the rods to communicate with other types of neurons in the retina.
When assessed in mice with congenital blindness, the Müller glia-derived rod photoreceptors were found to be functional as they could communicate with other retinal neurons across synapses.
Currently, the researchers are conducting behavioural studies to determine if the ability to perform visual tasks has been restored in the mice.
The team also intends to investigate the application of the new procedure on cultured human retinal tissue.




