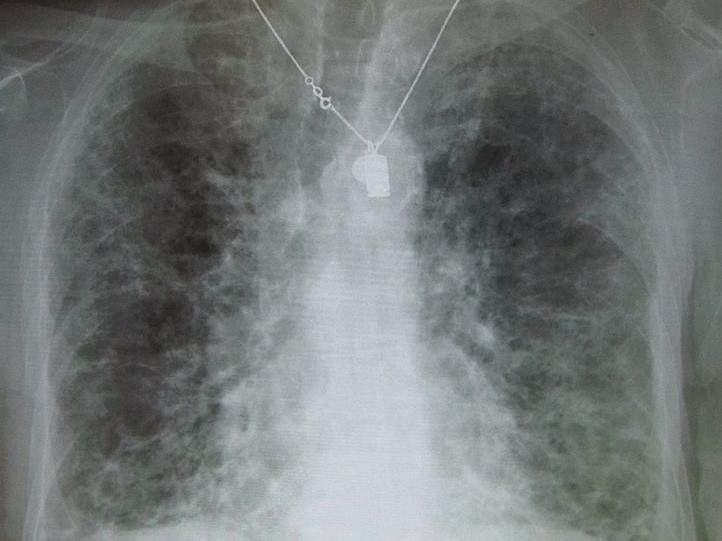
A research team from Helmholz Zentrum München’s Department of Lung Repair and Regeneration (LRR) with support from the University of Denver claim to have discovered the mechanisms underlying the pathological tissue changes that occur in patients with idiopathic pulmonary fibrosis (IPF).
According to their findings IPF is associated with higher numbers of extracellular vesicles that relay WNT5A signalling molecules to cells in the lungs.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
IPF is an aggressive form of pulmonary fibrosis, a disease associated with the increased formation of connective tissue in the lungs, which causes scarring (or fibrosis) of functional lung tissue and subsequently reduces the lung’s ability to take in oxygen and release carbon dioxide. Existing drugs are able to slow the progression of IPF, but cannot stop it permanently.
Previous research at Helmholtz Zentrum München by LRR and the Institute of Lung Biology and Disease concluded that the WNT5A signalling molecule is responsible for stimulating the proliferation of connective tissue cells in the lungs. This pushed further studies into the general WNT signalling pathway. However, scientists were unsure whether and how extracellular vesicles are involved in the transport of the WNT5A molecules.
Lead author of the LRR study Aina Martin-Medina said: “We were able to show in the study that increased levels of extracellular vesicles occur in IPF patients, which then act as carriers of WNT5A.”
The researchers also showed in petri dish experiments that by reducing the number of vesicles caused a decline in tissue scarring.
Another of the authors, Dr Mareike Lehmann, said: “Simply put, extracellular vesicles are tiny pouches released by cells that can contain a large number of messenger substances, such as proteins and nucleic acids. They are an important means of communication between cells and organs and help to ensure that the substances reach completely new sites.”
The LRR team now plans to examine the suitability of extracellular vesicles as a possible pharmacological biomarker and as a possible therapeutic target.




