Boehringer Ingelheim microParts, a subsidiary of Boehringer Ingelheim Group, invested $94m in a new atomisation factory in Dortmund as part of its expansion strategy. The facility, which was built at the company’s existing premises in Dortmund, was inaugurated in September 2010.
The new factory, which the company calls Module 2, produces Respimat Soft Mist Inhaler devices, which are sold under the brand names Spiriva and Berodual.
Respimat was co-developed by STEAG microParts, which was acquired by Boehringer Ingelheim in 2004. The device was launched in 2004 in Germany in combination with Berodual and is exclusively manufactured for the Boehringer Ingelheim Group of companies.
The expansion doubled annual production of inhalation devices, and at the same time created 150 additional jobs for full capacity production of Respimat.
In September 2014, Boehringer Ingelheim announced further plans for expansion at the site. It will invest €100m ($124m) to increase the annual production capacity to 44 million units. The expansion is expected to create 100 new jobs at the site
Project
A foundation stone for the new factory was laid in March 2007. Dortmund was the preferred site, due to the streamlined structure of the location and presence of an excellent local network.
Expansion increased the annual production of Respimat to approximately 20 million devices. The increased production ensures supply of Respimat in the global market for patients suffering from chronic obstructive pulmonary disease (COPD).
Facility
The atomisation factory is a four-storey building. Its facilities are spread over 12,000m², of which 4,000m² is dedicated as a clean room area.
Space for 150 new high-tech modern work stations was created for new employees.
A high stacker warehouse was built adjacent to the factory building to deal with increased material flow.
Construction
Construction required 2,000t of concrete and 1,300t of steel. A high-tech bubble ceiling was integrated to keep the production rooms column free. To reduce the weight of the ceiling, 25% of concrete was replaced with 1,600 plastic balls. The diameter of the balls is 44cm.
Foundations of the high-bay warehouse were dug 15m deep to facilitate necessary storage.
Two-thirds of the total investment was spent on technical equipment and production systems.
Respimat Soft Mist inhaler
Respimat Soft Mist inhaler is propellant-free and delivers a measured dose of medication in the form of fine mist.
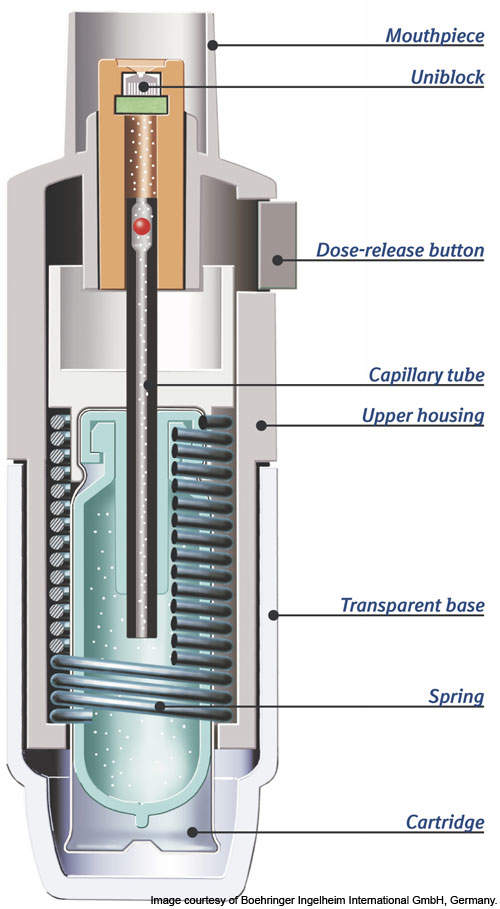
The device has a spring, cartridge, a transparent base, capillary tube, the dose-release button, dose indicator, dosing chamber and a fine nozzle system called uniblock.
The device is eco-friendly and does not require batteries. A spring placed in the inhaler’s base is compressed when twisted at 180° building up mechanical power that releases the solution through uniblock.
Solution is stored in a collapsible plastic bag, which serves as a reservoir. With each actuation the correct dosage is drawn from the plastic bag sealed in a plastic container inside the cartridge.
The exact dose is present in the dosing chamber. It is released when the dose-release button is pressed. The transparent base and capillary tube slide down allowing the dosage to flow through the micro pump.
A dose indicator on the device shows the number of doses left after each use. The red zone indicates that only 30 doses are left. The device automatically locks when the indicator reaches the end of the scale.
Uniblock is the most unique mechanism of the device. It is an extremely fine nozzle through which the medication is forced in the form of a fine mist. The liquid droplets are fine particles less than a fraction of 5.8µm average.
Marketing commentary
Boehringer Ingelheim microParts was established in 1990 as STEAG microParts. It was acquired by Boehringer Ingelheim Group in 2004.
Boehringer Ingelheim is one of the world’s 15 leading pharmaceutical companies. It develops, manufactures and sells micro-system-technology (MST) products for the life science industry.
It has expertise in areas such as inhaler technology, diagnostics and analytics, microfluidics and microoptic products manufactured from a wide variety of plastics and silicon.
The company’s activities include design development, prototype manufacturing, production and worldwide marketing. Some of the products they have available in the global market are propellant-free aerosol systems, micro-structured reaction platforms and micro-spectrometers.