Cordis Corporation was established in Miami in Florida, US in 1959. The company is a subsidiary of the pharmaceutical major Johnson & Johnson, producing medical device products (drug-eluting stents) for patients suffering from coronary artery disease and a variety of other vascular disorders.
In July 2006 Cordis, in conjunction with Johnson & Johnson, decided to make a €100m investment to secure the future of production in Ireland and create a new complement of 460 jobs over a three-year period.
This included 400 people in manufacturing and 60 additional positions at the finished goods testing centre and at the development laboratory.
The 180,000ft² plant at Cashel in County Tipperary, which combined the functions of manufacturing and development, was built on a 40-acre site adjacent to Alza (another subsidiary of Johnson & Johnson) and a producer of drug delivery systems.
Alza was built in 2001 and is a large-scale manufacturer of products incorporating D-TRANS transdermal technology, such as NicoDerm CQ and Durogesic.
Cordis at Cashel provided the entire production of drug eluting stents for Johnson & Johnson in all markets outside of the US. Alza and Cordis share some common infrastructure and facilities on the site, such as quality control and manufacturing services.
The plant, the sixth for Johnson & Johnson in Ireland, became operational in the first quarter of 2008. The investment was supported by the Irish Development Agency (IDA).
In June 2011, Johnson & Johnson announced the closure of the Cashel plant. The decision was part of a restructuring programme aimed towards cutting 1,000 jobs worldwide. The closure was also due to Cordis’ decision to cease manufacturing of its CYPHER drug-eluting stents.
Cordis Corporation and its pharmaceutical products
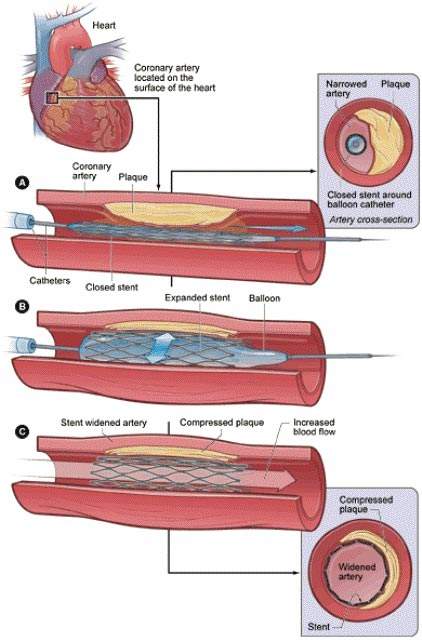
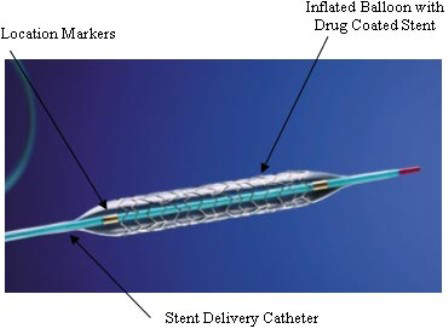
Cordis is one of the world leaders in producing drug-eluting stents (DES), which offer an effective alternative to open-heart surgery.
A drug-eluting stent is a coronary stent that can loosely be described as a supporting scaffold, which is placed into narrowed and diseased coronary arteries and slowly releases a specialised drug that will slow and prevent cell proliferation.
The use of the drug-eluting variety of stent prevents the process of fibrosis that, alongside clots formation, could block the stented artery (restenosis). The stent is usually placed in the coronary artery by an interventional cardiologist during an angioplasty procedure, which is preferable to full-blown open-heart surgery.
DES use is now much more preferable to bare metal stents because they have a higher success rate and much lower rates of subsequent major adverse cardiac events (MACE).
Stent and drug combinations
The Cashel facility used the latest state-of-the-art technologies for stent manufacture and developed future drug-device combinations in the field of interventional cardiology, with major process and product development capabilities. The facility includes an 180,000ft² production building along with a development laboratory and a finished goods testing facility.
The facilities supported demand for the CYPHER family of drug-eluting stents (launched in 2003), including the CYPHER Sirolimus-eluting Coronary Stent and the CYPHER SELECT Plus Sirolimus-eluting Coronary Stent.
The latter received the CE mark in June 2006 and became available outside the US in the third quarter of 2006.
Extended facilities at Cordis’ Cashel site
Following the opening of the Cordis facility at the start of 2008, an extension project was launched.
The project involved construction of a new quality control laboratory and an extension to the canteen at the site (the work was carried out near the cleanroom facilities).
Work included demolition and extension of the existing canteen and office space and construction of a new quality control laboratory and associated infrastructure.
The work on the canteen was carried out in three phases, two construction phases and a final demolition of the existing canteen to allow continued use of facilities during the construction phase.
The laboratory was a single-storey steel and concrete construction with a suspended concrete on metal deck roof to allow for a possible first floor in the future. The architect / engineer for the €5.7m project was PM Group and the builder and civil engineer was L & M Keating. This project was completed by December 2008.