Eli Lilly opened a facility in Concord, North Carolina, in June 2024 to manufacture parenteral (injectable) products and devices, with an investment of $2bn.
The facility is the company’s second manufacturing site in North Carolina after its $470m state-of-the-art injectable drug and device manufacturing facility in Research Triangle Park in Durham, North Carolina, US.
The expansion of Eli Lilly’s footprint in North Carolina was intended to support the continuous development of the company’s existing portfolio, as well as facilitate the production of its future medicines.
The facility generated 600 employment opportunities in the region.
Location
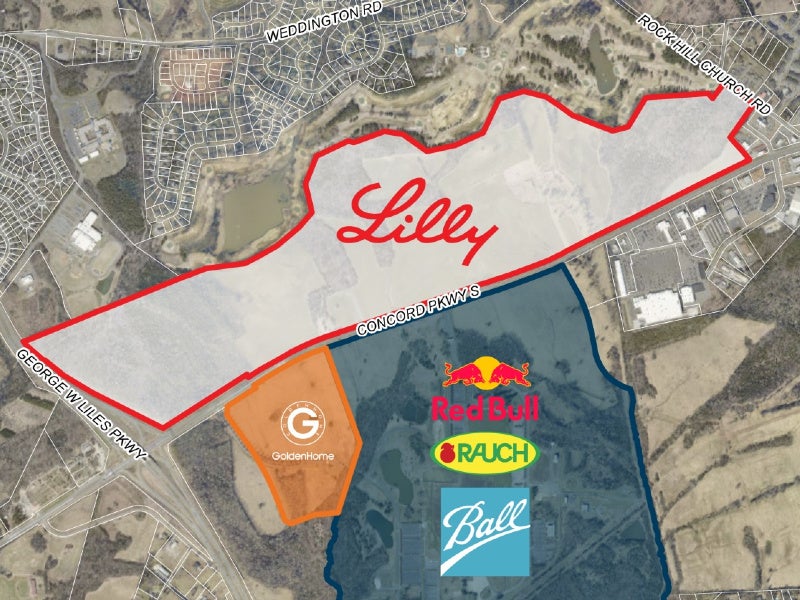
The manufacturing facility was built on a 400-acre (161.8ha) site at The Grounds at Concord, located in Concord city in Cabarrus County. It was built on the site of a former Philip Morris plant.
The site was selected due to its proximity to universities with strong science, technology, engineering, and mathematics programmes, its accessibility to major transportation infrastructure, and the availability of a local workforce with expertise in manufacturing technology.
Eli Lilly’s parenteral product and device manufacturing facility details
Eli Lilly’s state-of-the-art facility encompasses an area of 1.3 million square feet distributed among ten interlinked buildings.
The facility accommodated filling, packaging, automated storage and retrieval systems, as well as shipping and receiving operations.
Many administrative offices alongside quality assurance and analytical chemistry labs were also featured in the facility and there were dedicated areas for logistics and a central utility plant.
Economic incentives for the project
The project was partially supported by a 12-year Job Development Investment Grant approved by North Carolina’s Economic Investment Committee. It is expected to add $5.7bn to the state’s economy over the 12-year period.
The Cabarrus County and the City of Concord have offered performance-based property tax grants on the company’s total investment in the project, which were applicable based on the company’s ability to meet the operational performance goals.
Eli Lilly’s product portfolio
Eli Lilly offers medicines across a range of indications such as diabetes, cardiovascular, cancer, neurology, neurodegeneration, pain, bone muscle joint, endocrine and immunology.
Among the company’s approved therapies are Adcirca, an oral medication used for treating pulmonary arterial hypertension, Alimta, which is prescribed for the treatment of non-squamous non-small cell lung cancer, and Baqsimi, a prescription drug indicated to treat severe hypoglycaemia.
Amyvid is a radioactive imaging agent used to estimate β-amyloid neuritic plaque density in the brain of patients with Alzheimer’s disease and other causes of cognitive decline.
Forteo is a parathyroid hormone analogue medication used in the treatment of osteoporosis in high-risk postmenopausal women, as well as patients who have failed or are intolerant to other osteoporosis therapies.
The company has also developed Bamlanivimab, a Covid-19 vaccine authorised for emergency use by the US Food and Drug Administration.
Contractors involved
The construction companies D. A. Everett Construction Group and Gilbane Building Company, and construction engineering companies Bennett & Pless and V3 Companies were involved in the construction of the project.
WGM Design, an architecture company, LandDesign, a civil engineering and landscape architecture company, and Gensler, an architectural services provider, were responsible for the design and architecture of the facility.
SouthWood, a signage services provider, and Lithko Contracting, a concrete contractor, were also involved in the project.
Marketing commentary on Eli Lilly
Eli Lilly focuses on the development of high-quality medicines. The company was founded in 1876 by Colonel Eli Lilly and is based in Indianapolis, Indiana.
The company has more than 45,150 employees worldwide, with around 10,694 of employees engaged in research and development.
It has manufacturing plants in nine countries, conducts clinical research in more than 55 countries, and markets its products in about 105 nations worldwide.