Genentech is one of the most successful biotech companies on a world scale with four drugs prominent on the commercial market and around 50 drugs in its pipeline. The company is headquartered in South San Francisco, US, and has one of its major production centres at Vacaville in California, 54 miles northeast of San Francisco.
The 427,000ft² Vacaville facility came into operation in 1998 after being licensed by the FDA in 2000. It is situated on a 96 acre site, purchased in the late 1990s and has 12 x 12,000l fermenters in seven buildings on the site.
The facility employs 940 personnel. This was Cell Culture Plant No 1 (CCP 1).
Decision
In 2004 Genentech made a decision to expand and invest around $1bn in a new facility. At the time, the company had four products either in or coming into commercial production and it needed the additional capacity.
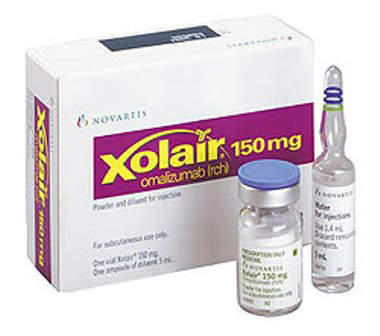
The four products are rituximab (branded Rituxan), trastuzumab (branded Herceptin), omalizumab (branded Xolair) and bevacizumab (branded Avastin). All are approved by the FDA.
The announcement of the new investment was met with a flurry of offers from a number of states in the US where there were suitable sites. However, the final decision to stick with the Vacaville plant was taken for three major reasons; the site already had space for a new plant on the 96 acre site, the majority of the necessary infrastructure was already installed; and Genentech wanted to get the plant into operation as quickly as possible and the best chance was to expand at Vacaville.
Expansion
The ground breaking for the expansion, called CCP2 (Cell Culture Plant 2), occurred in the third quarter of 2004. The expansion included a new facility on the same site consisting of three buildings (380,000ft²) on 20 acres north of the existing facility.
The 450,000ft² CCP2 was completed in 2009 and licensed by the FDA in the same year. It has eight 25,000l fermentation vessels.
Contractors
The general contractor for the expansion was DPR Construction Inc of Redwood City. The facility was completed and substantially handed over to Genentech in June 2007.
As well as the CCP2 buildings the project has also seen an expansion of the existing warehouse space and the central utility plant was doubled in size. Fluor Corporation was the architect for the CCP2 building.
Honeywell
In May 2006 Honeywell was selected to supply the integrated manufacturing control system for the new CCP2 expansion. The control system was designed to give consistency in production and also to provide a single paperless electronic record of operations.
The system integrates the Experion Process Knowledge System (PKS) and the POMSnet Manufacturing Execution System (MES) to support the plant's operational efficiency, providing electronic records of each batch of drugs produced to help reduce operator errors.
The operations system also features TotalPlant Batch and Uniformance Process History to enable the more efficient collection storage and analysis of data. POMSnet complies with 21 CFR Part 11, as well as ISA S-88 and S-95 manufacturing standards.
The system not only verifies that the equipment and materials used in processes are correct, but also checks their quality and availability.