Grindeks is one of the leading pharmaceutical companies in the Baltic States and its main areas of activity are heart and cardiovascular, CNS and anti-cancer medication.
The company is active in R&D and the manufacturing of original products, generics and active pharmaceutical ingredients (APIs). The Grindeks Group consists of four subsidiary companies in Latvia, Estonia and Russia, and exports 96% of its products to 40 different countries.
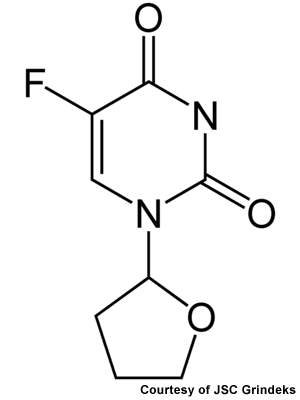
Some of the company’s best-known drugs are Mildronate, which inhibits the activity of gamma-butyrobetain-hydroxylase, causing a decrease of carnitine biosynthesis and is used in the treatment of angina, and Ftorafur, which is an important anti-cancer medication used in the treatment of malignant tumours.
In 2008, Grindeks’ turnover for APIs hit LVL5.1m. The company regularly manufactures 16 types of complicated APIs created in multi-stage synthesis. Its main products are Mildronate, Ftorafur (tegafur), zopiclone, oxytocin and a group of special veterinary APIs.
Highlighting the company’s focus on expansion for final dosage forms and APIs in August 2009, Grindeks announced a new active pharmaceutical ingredient, ursodeoxycholic acid. This can be used in the treatment of hepatic and gallstone diseases, a new therapeutic area for the company.
New Riga plant
In January 2009 Grindeks opened its new final dosage forms plant in Riga, Latvia; the plant was opened by the president of the country, Valdis Zatlers. This new plant, with a total floor space of 5,500m², was designed to meet all cGMP requirements and will be one of the most modern in the Baltic States.
The project required an investment of LVL9.1m ($16.8m) over the two years of construction, of which LVL6.4m was required for the new building and LVL2.7m was needed for new technology and equipment to outfit the plant.
The new plant was designed and the construction project managed by LSMW of Poland. The construction work was carried out by the Latvian construction company RBSSkals, with Bosch providing some of the equipment.
Production and manufacturing
The plant has been designed with one of the most modern automatic control systems possible; this is able to check the microclimate in the premises, operate the engineering systems, control production processes and ration energy resources for economy.
It is expected that the new facility will increase the company’s manufacturing capacity to 1.5 billion tablets and 500 million capsules per year. This will help to meet the increase in demand for products which is currently approaching 20% per year. In addition, the facility will employ a total workforce of 102 employees who will be production operators, engineering technicians, pharmacists and chemists.
Finance
The new plant was financed by funds from a 2005 share issue and also a bank loan of around LVL2.5m. This was one of the company’s largest investments in its history.
The long-term loan for the construction of the new facility was made by Swedbank (previously de jure JSC Hansabanka), which has had dealings with Grindeks for almost ten years.
The investment of more than LVL9m is expected to help increase the company’s competitiveness in European and world markets, according to Kirovs Lipmans, chairman of the Council of Grindeks.
The new world-class pharmaceutical plant is expected to provide great yield not only to the company, but also to the competitiveness of Latvia, encourage improvement of the national economy and promote an increase in the country’s exports.
Grindeks is now examining the feasibility of establishing a new facility to produce injectable (parenteral) drugs in the free port of Riga.