No Filter Selected
No Filter Selected
No Filter Selected
No Filter Selected
Cremer CFS-622x3 / CFS-622x4 Modular Counting System
Cremer has pioneered, developed and introduced multi-channel electronic counters for the pharmaceutical industry 30 years ago. Today, the counting principles...
Vertex’s Kalydeco obtains FDA approval to treat CF in children
Vertex Pharmaceuticals has received approval from the US Food and Drug Administration (FDA) for its Kalydeco (ivacaftor) to treat cystic fibrosis (CF) in children aged between two and five years.
Vertex submits NDA to FDA for triple combination CF drug
Boston-based Vertex Pharmaceuticals has announced it has submitted a new drug application (NDA) to the US Food and Drug Administration (FDA) for its triple combination therapy for cystic fibrosis (CF), VX-445 (elexacaftor), tezacaftor and ivacaftor.

Scotland reaches access agreement with Vertex for CF drug Orkambi
Vertex Pharmaceuticals has announced two of its cystic fibrosis (CF) drugs, Orkambi (lumacaftor-ivacaftor) and Symkevi (tezacaftor/ivacaftor) will now be available to eligible patients in Scotland through the National Health Service (NHS).
Vertex obtains EC approval for combination therapy to treat CF
Kaftrio in combination with ivacaftor will help enhance the quantity and function of the CFTR protein at the cell surface.
NHS England and Vertex break stalemate over price of CF drugs
Four years after Orkambi was approved in the European Union, this life-saving drug will now be available to Cystic Fibrosis (CF) patients in England through the NHS. The reason for the delay was continued failing pricing negotiations between Orkambi's manufacturer, Vertex, and NHS England, leading to patient advocacy campaigns supported by Labour Party leader Jeremy Corbyn.

Vertex gains FDA approval for expanded use of CF drug Trikafta
The expanded FDA approval was supported by an open-label 24-week Phase III trial.
Pipeline power: Sionna’s AbbVie deal could redefine CF therapeutics
The acquisition of AbbVie’s clinical compounds enhances Sionna’s ability to address these challenges and solidifies its position as a potential future player in the CF market.
FDA expands label of Vertex’s CF drug Symdeko to include younger children
Vertex Pharmaceuticals has announced that the US Food and Drug Administration (FDA) has expanded its approval of Symdeko (tezacaftor/ivacaftor and ivacaftor) to include cystic fibrosis (CF) patients aged six to 11 who have either two copies of the F508del mutation in the cystic fibrosis transmembrane conductance regulator (CFTR) gene or one mutation in the CFTR responsive to the drug.

4DMT’s CF gene therapy posts early cues for improving patient outcomes
The company presented the Phase I/II data at the ECFS meeting, with improved outcomes in three cystic fibrosis patients.
ATS 2025: ETI-linked mucus clearance tied to lung function gains in paediatric CF
The prospective study enrolled 30 paediatric CF patients who underwent imaging and spirometry both before and following the initiation of CFTR modulator therapy.
ATS 2025: Real-world data supports Trikafta as gold standard in long-term CF care
The published long-term data suggests that the treatment could maintain its dominant position in the CF market.
FDA rejects Vertex's sNDA for Kalydeco to treat cystic fibrosis in people aged two years and older
The US Food and Drug Administration (FDA) has rejected Vertex Pharmaceuticals' supplemental new drug application (sNDA) for Kalydeco (ivacaftor) designed to treat people with cystic fibrosis (CF) aged two and older, with one of 23 residual function mutations in the CF transmembrane conductance regulator (CFTR) gene.

Marking milestones at the North American Cystic Fibrosis Conference 2017
The 2017 North American Cystic Fibrosis Conference (NACFC) was held at the Indiana Convention Center in Indianapolis, Indiana from November 2–4.
Current and future players in the cystic fibrosis market
Few pipeline agents in late-stage development mean the competitive positions of CF market players are unlikely to change.
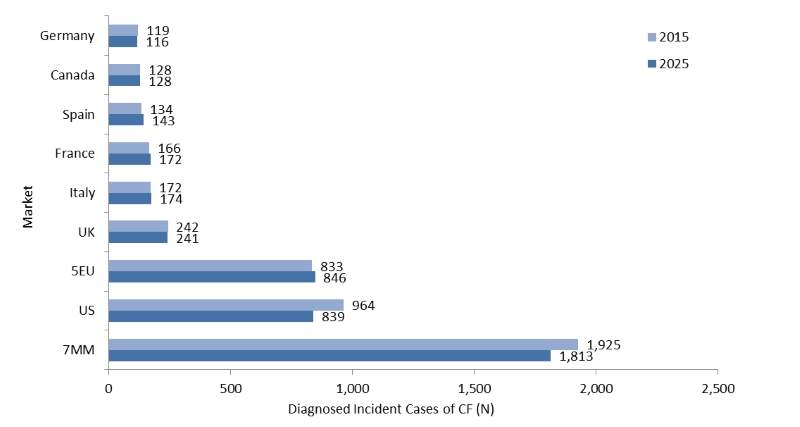
Diagnosed incident cases of cystic fibrosis are set to decrease
The diagnosed incident cases of cystic fibrosis (CF) are expected to decrease between 2015 and 2025 in the seven major markets (7MM) of the US, France, Germany, Italy, Spain, the UK, and Canada.
Vertex’s Trikafta: treating the genetic basis of cystic fibrosis
At the end of 2019, the FDA approved Vertex’s Trikafta, the world’s first triple combination therapy for cystic fibrosis. This drug’s mechanism of action means it treats the underlying genetic cause of the disease in 90% of the CF population. Allie Nawrat investigates the benefit this drug brings to at-need CF patients and how to treat the 10% of patients who remain without effective treatment.

Cystic fibrosis: matching medicines with mutations
The 2017 North American Cystic Fibrosis Conference (NACFC) was held at the Indiana Convention Center in Indianapolis, Indiana from November 2–4. The conference annually brings together scientists, clinicians, and caregivers worldwide and facilitates discussion about ways to improve the health and quality of life for cystic fibrosis (CF) patients. Sponsored by the Cystic Fibrosis Foundation (CFF), the 2017 NACFC featured a wide variety of session formats, including didactic symposiums, interactive workshop sessions, brown bag luncheons, and grand rounds. Each day of the conference was highlighted by a themed plenary session headed by leading experts in the CF community. This piece will discuss the significant advances made by the pharmaceutical industry in addressing the unmet needs of CF patients.
Vertex to acquire Concert Pharmaceuticals’ cystic fibrosis drug for $250m
US-based Vertex Pharmaceuticals has signed an agreement to acquire CTP-656, an investigational cystic fibrosis transmembrane conductance regulator (CFTR) potentiator from Concert Pharmaceuticals for up to $250m.
Trikafta/Kaftrio continues to be a game-changer for cystic fibrosis patients
Vertex Pharmaceuticals’ Trikafta/Kaftrio is currently the world’s first triple-combination cystic fibrosis therapy.
