
More labels to manage, changes requested, expansion into new markets, more and more versions of files to track; does this sound familiar?
How about lack of visibility into your E2E labeling process or logging in and out of multiple systems each day to manage labeling information?
Gaining visibility into the E2E labeling process is paramount. Without it, it’s difficult to make smart business decisions when changes are required, resulting in loss of productivity.
Let’s go behind the scenes of E2E Labeling and discuss a typical current state for the process, how it’s improving, and what the future may hold. Read on to learn some tips and tricks to help you start improving your current process.
The Current State of E2E Labeling
What factors make gaining visibility into the E2E labeling process a challenge? There are many, including:
- The volume of content from Company Core Data Sheets (CCDS), Patient and Product Information, Instructions for Use (IFUs) or User Guides, to the label or carton
- The relationships between a change request, the impacted CCDSs, the local market impact, and the SKUs and components affected
- The number of people involved in the process, from quality, regulatory, and artwork design to supply chain, potentially in multiple countries or regions
- The complex technology infrastructure that supports the E2E labeling process
E2E labeling is different for every organization. However, it’s common for companies to use the following E2E process:
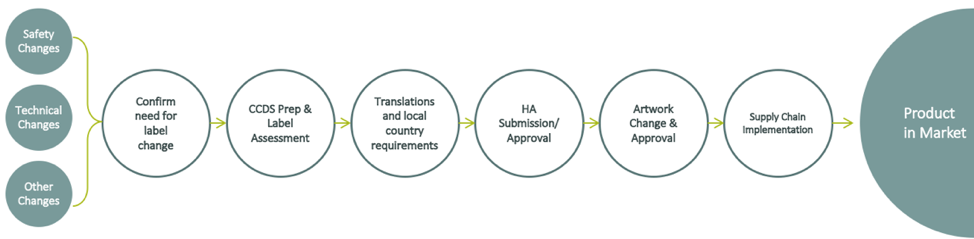
Oftentimes, companies find issues with:
- Change Control
- Product Registration
- Document Management
- Bill of Materials Management
- Specification Management
- Health Authority Submission
- Regulatory Information Management
- Artwork Management
- Supply Chain Management
A company may have more than one of each of these systems, either due to regional differences or legacy systems from an acquisition. Or it could be because there is decentralized sponsorship of operations in the business; it’s not unusual to see upwards of 20 instances/versions of SAP running in a global organization.
With this complex supporting infrastructure, when someone in the company wants consistent E2E visibility, there is no one place to get it. Then it becomes a manual effort to populate Excel spreadsheets with data collected from multiple systems. Manually filling and managing spreadsheets is an enormous effort and as soon as it is created, it is often outdated. With this type of knowledge, many companies realize the need to improve the current state with appropriate technologies.
The Evolving E2E Technology Landscape
Some companies may have already started the journey with system consolidation or integrations. Solution providers are continuing to improve their offerings to meet this growing demand for better visibility. The following three categories describe what Esko is currently seeing in the marketplace:
- Global System Standards
- Integrations
- Automation
Global System Standards
Global system standards (GSS) are driven by global process harmonization, and for the most part, life sciences companies are pretty good at implementing GSS.
Years ago, regions may have selected different tools for the same function because the tools were not mature enough to handle regional variations. Now, however, most enterprise applications accommodate global usage. But even with a consistent global infrastructure, when an acquisition takes place, there are different SOPs and supporting technologies that need to be combined and rationalized.
E2E visibility implies that a company has access to this data, with the ability analyze it and report results. Data Warehousing, a common reporting approach, lets users create real-time bar charts and other graphical views of the data. Warehousing requires data modeling and “denormalizing” data, effectively organizing the data so users can easily get the metrics they need.
Denormalization has both positives and negatives. Data warehousing dramatically reduces the end user’s need to understand complex data models from various source systems because the data is structured, or “denormalized.” However, the data models are built based on what types of reports you want today. If your future needs change, it may require a change to the data model to accommodate the new requirements. Some companies use Data Lakes to address this challenge.
Think of a Data Lake as a holding area for the raw data from each system used in the E2E labeling process. With a Data Lake, you don’t need to know what reports you’ll want in the future; since there is no denormalization of the data, it can be more easily manipulated to meet your future requirements, whatever they may be. There is a lot more data to manage with a Data Lake model, but the tools used to analyze and report on data are more equipped to handle the increased volume.
Integrations
Years ago, most integrations required custom code. Now, there are industry standard tools like Web Services, XML and APIs to simplify connectivity. Integrations are continually improving via “connectors” that eliminate the need for any customization between applications. Common data models and taxonomy are a critical factor in a seamless integration model as well. If attributes are labeled the same way throughout the E2E systems, it dramatically simplifies the connectivity design.
Having a set of systems that seamlessly exchange information not only improves data quality and eliminates duplicate data entry, but it also gets companies closer to true E2E visibility.
Single Sign-On (SSO) and portals are increasing in popularity. SSO enables companies’ employees use the same credentials to log in to the various E2E systems. With portals, users don’t have to remember or bookmark the URLs of the systems they work in. A user’s portal controls their task list, and the portal manages access to the underlying systems.
Automation
There are many examples of automation in a typical current state E2E labeling process. For example, an event occurring in one system can automatically trigger an event or activity in another system. It could be as simple as a change request e-form routing the request to the right people based on the type of change requested. It could be a quality check, such as an automated proof-read being done on a file when it’s uploaded. There are lots of examples of automation used today; however, there is much more that could be done.
Many companies are now doing some level of content management but still rely on document-centric data, vs. database-centric data. By defining ‘content elements,’ whether they are images, logos, barcodes or copy, a company molecularizes the traditional digital asset allowing organizations to reuse the elements. They can also perform impact analyses of a change quickly and nimbly when rolling out changes. This molecularization builds the foundation needed for companies to move from automation to intelligence.
Consider an example of a change to a warning statement in the future: When entering a change request, the appropriate person receives a notification of the CCDSs impacted, and any affected regions are also automatically notified. The change request event automatically kicks off the required processes for the labeling change (IFU, User Guide, label, carton, etc.), includes the right people in the process, and highlights the exact text that must be changed. It can even replace the old warning statement with the new one automatically.
Furthermore, the user is notified that some of the components needing to be changed are already routing due to another change. The notification also provides recommendations on bundling/unbundling based on where the components are in the approval process, how important the new change is, and how much inventory is available.
Companies must build the foundation of process and system standards, along with the integration of the systems that support E2E labeling, and molecularization of content, to get to this future “intelligent” state. It won’t happen overnight, but digital tools help companies discover real E2E visibility by driving efficiencies and quality through automation and intelligence.
Future State
Below is an example of what a company’s future state could look like. Using a single Data Lake and E2E globally integrated systems, a company could gain much clearer visibility into their entire labeling process.
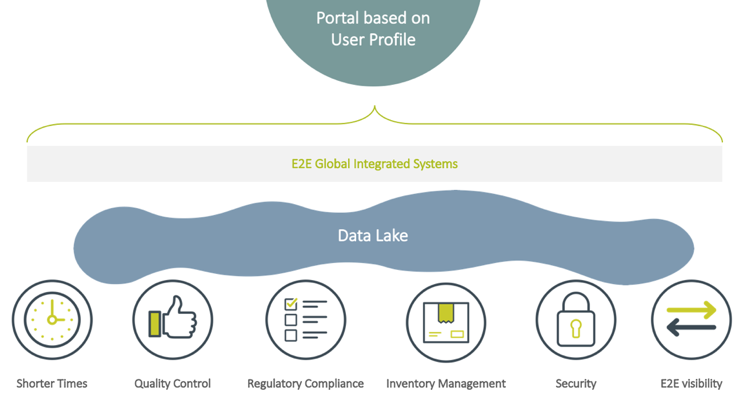
There are many benefits to E2E visibility – companies get their products to market faster, improve product packaging quality, and minimize the risk of errors (which means fewer recalls), and consistently adhere to regulatory standards. Users collaborate across departments, removing the miscommunication (or lack of communication) that occurs between operational silos. Inventory management is easier because users can log-in to the appropriate integrated system and analyze data from the Data Lake. And using SSO, a user can access all the systems from a secure portal.
Summary and Next Steps
While the E2E process is similar from one company to the next, each company has a different combination of systems that support and manage labeling and artwork. If you’re putting a plan together, whether it’s focused on building the foundation, or introducing automation, here are some tips to get you started:
- Be open to change
- Identify areas of risk and opportunity. If you’re part of the E2E labeling process, you likely see examples of this every day
- Use a Digital Maturity Model to help you determine your current state and define your future state
- Begin implementing changes to your current state; small wins go a long way in your overall plan
- Select technology partners that have integration capabilities, experience, and an understanding of the E2E labeling process and its complexities.
To learn more about how Esko can help you with your E2E visibility, contact us.


