Powering the Organoid Revolution with Scalable, Human-Based Systems
The strategic shift to 3D biology
Organoids, or miniature organ-like structures derived from stem cells, have emerged as transformative tools in biomedical research. Unlike traditional two-dimensional (2D) cell cultures, organoids possess a three-dimensional (3D) architecture and multiple cell types, closely mimicking the structural and functional properties of native organs. This makes them highly versatile, scalable platforms for studying human biology in a controlled laboratory environment.
As regulatory landscapes evolve, the demand for these models is surging. In April 2025, the US Food and Drug Administration (FDA) announced plans to phase out certain animal-testing requirements, explicitly promoting organoids as superior, human-relevant alternatives for drug development. With over 180 organoid-related clinical studies registered by mid-2025, the industry is rapidly transitioning toward these high-fidelity systems for drug sensitivity testing and precision medicine.
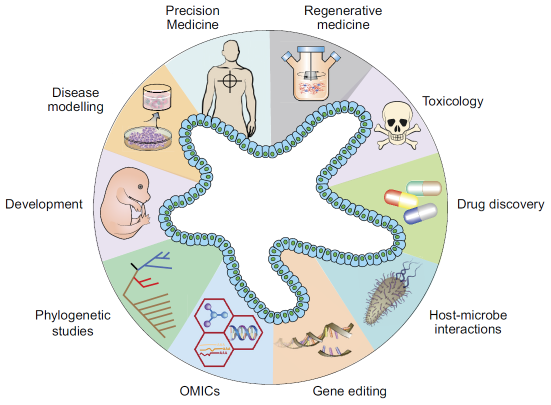
Figure 1. Diverse applications of organoid technology1. (Image from Corrò et al. used under CC BY 4.0. https://doi.org/10.1152/ajpcell.00120.2020)
Technical hurdles in organoid culture
Despite their potential, the path from bench to bedside is hindered by persistent technical challenges:
- Batch-to-batch variability that compromises experimental reproducibility.
- Insufficient tissue maturation and limited scalability.
- Restricted access to clinically compatible, high-performance reagents.
- Endotoxin contamination, which can activate innate immune signalling, alter cellular metabolism, and induce stress responses, ultimately confounds drug response data.
Sino Biological’s 3D organoid research solutions
To address these bottlenecks, Sino Biological offers a comprehensive portfolio of high-quality cytokines, growth factors, small molecules, and marker antibodies that are essential for organoid culture and characterisation. Our research solutions are optimised for nearly 30 popular physiological and tumour organoid models, providing robust support for oncology, disease modelling, and drug screening.
For example, Fudan University’s Jianqing Liang, PhD, and his colleagues, used Sino Biological’s recombinant human EGF protein (Cat#: 10605-HNAE) to establish an optimal culture system for human fetal thyroid organoids (hFTOs)2. Figure 2A depicts the schematic diagram of the digestion and seeding of fetal thyrocytes, and the culture medium of hFTOs, while Figure 2B represents the time point bright field images of hFTOs.
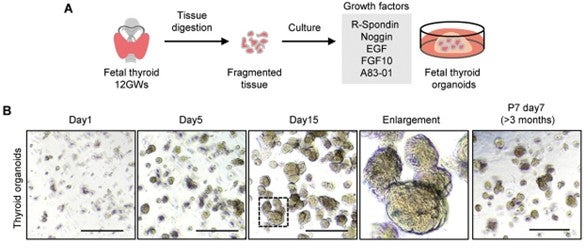
Figure 2. Generation of reproducible human fetal thyroid organoids (hFTOs)2. (Image from Liang et al. used under CC BY 4.0. https://doi.org/10.1002/advs.202105568)
To further support the scientific community, Sino Biological offers a consolidated hub of technical guidelines, brochures, and expert-led webinars designed to streamline your organoid research workflow.
The ProPure™ advantage: ensuring culture integrity
In organoid culture systems, recombinant cytokines and growth factors play central roles in directing stem cell differentiation, maintaining tissue identity, and sustaining long-term growth. However, trace levels of endotoxin contamination can profoundly disrupt three-dimensional cultures by activating innate immune signalling pathways, altering cellular metabolism, and inducing stress responses or cell death. Such unintended effects compromise experimental reproducibility, obscure true disease phenotypes, and confound drug response data, particularly in sensitive, patient-derived organoid models.
Sino Biological provides ProPure™ endotoxin-free proteins for researchers to establish robust and translationally relevant organoid platforms. Highly purified preparations minimise inflammatory artefacts, reduce batch-to-batch variability, and enable consistent lineage specification across large-scale cultures. In high-throughput drug screening and preclinical studies, these reagents help ensure that observed biological responses arise from therapeutic candidates rather than culture-induced artefacts, supporting confident data interpretation and regulatory-facing research workflows.
Conclusion
Organoid technology is reshaping disease modelling and drug discovery by providing physiologically relevant, human-based platforms. Overcoming challenges such as variability, limited scalability, and endotoxin interference is essential to fully realise their translational potential. With high-quality, endotoxin-free reagents and comprehensive technical support, Sino Biological empowers researchers to build robust, reproducible organoid systems that accelerate scientific innovation and therapeutic development.
Learn more at www.sinobiological.com
References
- Corrò C, Novellasdemunt L, Li VSW. A brief history of organoids. Am J Physiol Cell Physiol. 2020;319(1):C151-C165. doi:10.1152/ajpcell.00120.2020
- Liang J, Qian J, Yang L, et al. Modeling Human Thyroid Development by Fetal Tissue-Derived Organoid Culture. Adv Sci (Weinh). 2022;9(9):e2105568. doi:10.1002/advs.202105568