Clinical trial numbers for CAR-T therapies have risen dramatically in recent years, and as these programmes expand beyond oncology into larger, chronic indications, the industry is discovering a hard constraint: leukapheresis is becoming a structural bottleneck.
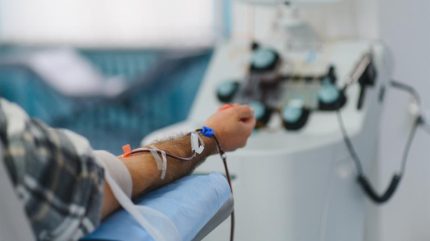
Leukapheresis is a specialised blood collection and separation procedure that selectively removes white blood cells, typically to collect immune cells such as lymphocytes for advanced therapies like CAR‑T. Blood is circulated through a machine that separates out the required cells, with the rest returned to the patient over several hours. However, the sector’s historically treatment centre-dependent collection model now sits at the heart of whether programmes can scale, launch on time, and expand access to diverse patient populations.
In a recent webinar, Future Proofing Cell & Gene Therapy: Strategies to Secure Your Leukapheresis Supply Chain, Heather Munoz, Principal of Global Market Development, and James Johnson, Director of Starting Materials and Research at BBG Advanced Therapies (a subsidiary of BioBridge Global, based in Texas), discuss today’s most persistent bottlenecks in sourcing well-qualified leukapheresis providers as part of autologous and allogeneic therapy supply chains.
Host Jack Cuthbertson, Senior Healthcare Analyst at GlobalData, opens by framing the scale and direction of travel in cell therapy, which is currently dominated by oncology indications. The data highlights a key structural shift: early‑stage pipelines are already skewing towards allogeneic ‘off‑the‑shelf’ approaches – a trend that will magnify pressure on starting material supply chains.
Leukapheresis as a structural bottleneck
Heather Munoz, drawing on her background in apheresis nursing and autologous CAR‑T, starts the discussion by pointing to a fundamental mismatch between demand and current infrastructure. “Quality variability and capacity constraints are the biggest challenges,” she says. “We’re starting to see more autologous CAR-Ts, more clinical trials, and we’re expanding into the much larger autoimmune population. We simply don’t have enough chairs to do apheresis or infusions.”
With next‑generation therapies moving into autoimmune, cardiovascular and respiratory indications, patient numbers are expected to rise sharply, yet most procedures still funnel through a small number of metropolitan academic centres, specialised in haematology-oncology. “We haven’t really gone into the community setting quite yet,” notes Munoz, “there’s a high volume that are going through academic centres, and it’s causing a lot of competition for which CAR-T therapies are going to hit the market.”
For many US patients, especially in rural or underserved regions, this comes at a significant personal cost: “They’re travelling two-plus hours to get to the facility to have an apheresis procedure done, which can take anywhere between four and six hours,” says Munoz. These burdens are often not reimbursed, directly affecting enrolment, retention and vein‑to‑vein time.
Munoz also highlights the fragility of heavily pre‑treated autologous patients: “This might be a line three treatment for them, so they’ve already gotten chemotherapy – which ultimately can impact the starting material with low T cell fitness and lymphocyte counts.”
When it comes to allogeneic therapies, re-using the same donors is also problematic, says Johnson, as the same donor doesn’t produce the same product every time. “There’s a lot of conversations in this space around super donors or recallable donors, but an over reliance on a single donor can present potential pitfalls as too much of the programme is hinged on their availability and persistent eligibility,” cautions Johnson.
Rethinking vendor strategies for scalable leukapheresis
Munoz describes frequent misalignment between collection sites and manufacturing expectations and argues for harmonisation of best practices and clearer standards for starting material quality. Johnson agrees and calls for stronger feedback loops between collectors, manufacturers and developers to anticipate roadblocks rather than react to failures.
Munoz also outlines what robust vendor qualification should include: clear definition of fit‑for‑purpose, mature quality systems (including FACT/AABB accreditation), standardised operating procedures, proven capacity and contingency planning, and reliable logistics for chain of identity and chain of custody.
A recurring theme of the webinar was that sponsors must treat leukapheresis centres as strategic partners from the earliest phases, not as late‑stage operational bolt‑ons. Johnson stresses: “It’s never too early to start planning and then implement when it’s appropriate to the phase.”
Both speakers emphasise the importance of deeper collaboration. Johnson notes that long‑term success depends on “the ability and the willingness to sit down together and collaborate.” Structured governance, regular joint reviews and data‑driven feedback on manufacturing outcomes were highlighted as critical to reducing variability, strengthening chain‑of‑custody controls, and ensuring that process improvements are implemented consistently across sites as programmes scale.
Mobile leukapheresis: bridging capacity and access
A highlight of the webinar was a discussion around the BBG Advanced Therapies mobile leukapheresis centre, positioned as a flexible way to support both clinical and commercial demand. Mobile units can provide surge capacity when academic centres are saturated, accelerate early launches while fixed sites come online, and extend access into rural areas, improving trial diversity and real‑world representativeness.
Johnson notes that, from a regulatory perspective, the mobile unit is designed as a like‑for‑like GMP environment with redundant power, environmental monitoring, electronic quality management systems and validated processes stress‑tested in real‑world conditions: “Anything that is necessary in a traditional brick and mortar site is going to be necessary in the mobile setting as well.” A single, centrally governed mobile platform can also simplify variability control and regulatory oversight by operating under one quality management system across multiple locations.
By treating leukapheresis as a strategic asset, and investing in robust partners, resilient networks and innovative models like mobile collection, sponsors can turn today’s patient access challenge into a competitive advantage for the next wave of cell and gene therapies.
To explore further – including additional detail on regulatory expectations, vendor qualification and chain‑of‑custody controls – download and watch the full webinar here.


