Arbovirus E Dimer Vaccines: Engineering Safer Immunogens to Circumvent Antibody-Dependent Enhancement
Current worldwide circulation and burden of Dengue and Zika
Arboviruses, particularly Dengue virus (DENV) and Zika virus (ZIKV), represent a massive and escalating global health threat. Transmitted primarily by infected Aedes mosquitoes, Dengue is now endemic in over 100 countries. It puts roughly half of the global population at risk, causing an estimated 400 million infections, 100 million symptomatic cases, and up to 40,000 deaths annually.
Zika virus, which triggered a global public health emergency during the 2015–2016 epidemic, continues to circulate widely (Figure 1). While often asymptomatic, ZIKV poses a devastating risk during pregnancy, leading to Congenital Zika Syndrome (CZS)—which includes severe microcephaly—as well as neurological complications like Guillain-Barré syndrome in adults. The geographic footprints of both viruses are rapidly expanding due to urbanisation, global travel, and climate change, underscoring the critical and immediate need for safe, globally deployable vaccines.
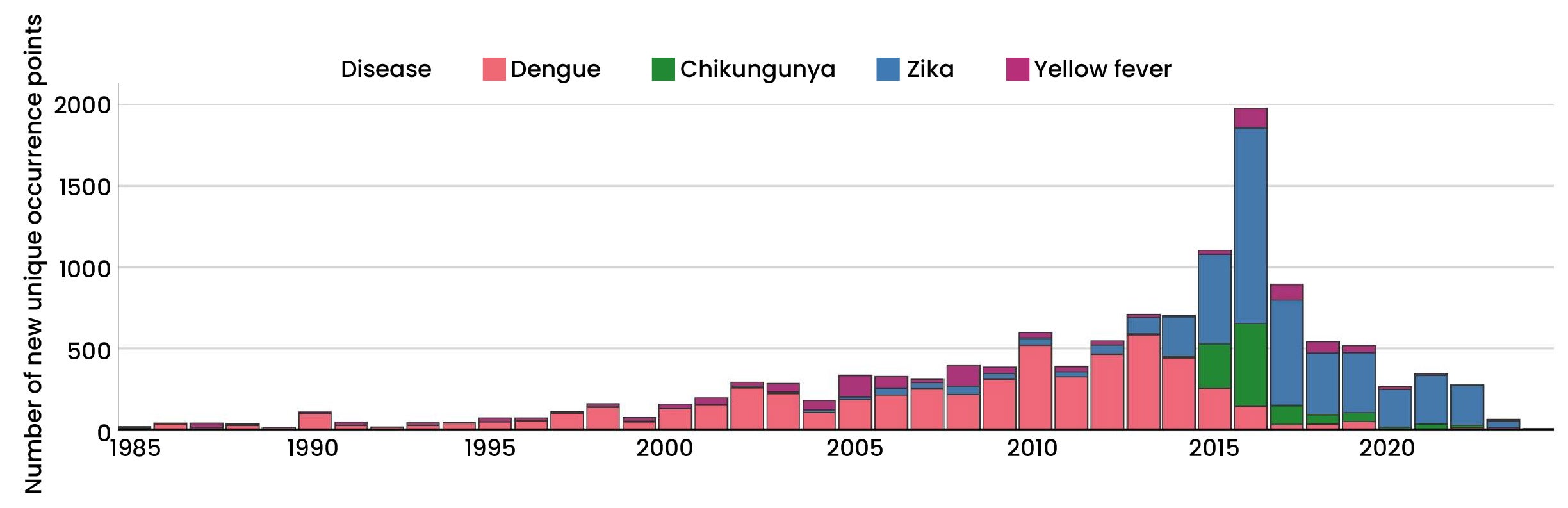
Figure 1. The global number of new unique occurrence points added each year for arboviruses. (Image source: https://pmc.ncbi.nlm.nih.gov/articles/PMC11986131/)
Clinical research status of arbovirus vaccines and the ADE challenge
The clinical development of vaccines against DENV and ZIKV has historically been complicated by a severe immunological hurdle: antibody-dependent enhancement (ADE). Because Dengue exists as four distinct, co-circulating serotypes (DENV1–4), and because DENV and ZIKV share significant structural homology, previous infections or poorly designed vaccines can induce cross-reactive, sub-neutralising antibodies.
These antibodies often target the highly conserved, immunodominant ‘fusion loop’ of the viral Envelope (E) monomer. Instead of neutralising the virus, these antibodies facilitate viral entry into host immune cells, paradoxically worsening disease severity upon subsequent infection. Consequently, modern clinical research has shifted away from simply inducing broad antibody responses toward highly targeted, structure-guided approaches.
To avoid ADE, next-generation subunit and mRNA vaccines are heavily focused on utilising stabilised Envelope (E) protein dimers. By locking the E protein in its native, dimeric conformation, researchers can effectively hide the dangerous, ADE-prone fusion loop while safely exposing highly potent, dimer-dependent epitopes that drive robust and safe neutralising immunity.
While early-generation live-attenuated platforms currently dominate the late-stage pipeline, the ongoing threat of ADE has spurred a diverse array of advanced vaccine candidates with recombinant E dimers in active clinical trials:
Table 1. Examples of Recombinant E Protein Dimer-Based Arbovirus Vaccine
| Developer | Vaccine Candidate | Target Virus | Platform Technology | Latest Development Status |
| Merck / Hawaii Biotech | V180 (DEN-80E) | DENV | Recombinant Antigen Subunit | Phase 1 Completed |
| VBI Vaccines | VBI-2501 | ZIKV | Enveloped VLP | Phase 1/2a Completed |
| Univ. of Oxford / UNC | Covalently locked dimer (cvD) vaccines | DENV/ZIKV | Recombinant Antigen Subunit | Preclinical / Near-Clinical: Complete |
Research applications
To support this critical shift toward structure-guided antigen design, Sino Biological offers a comprehensive portfolio of high-quality recombinant Arbovirus envelope (E) proteins, including highly stable E dimers.
Engineered to meet the rigorous demands of advanced vaccine research, these E dimers accurately preserve the native-like dimeric conformation, as validated by SEC-MALS. Whether you are designing safer subunit vaccines, developing multivalent nanoparticle displays, or building multiplex immune-profiling assays to isolate specific antibody subsets, Sino Biological’s recombinant DENV and ZIKV E dimers provide the reliable, highly pure antigens needed for success.