
In the first part of our Q&A with Grace Breen, SVP, Quality, Sharp, we explored valuable insights derived from her longstanding career in pharmaceutical quality leadership. The interview focused on the importance of building a proactive quality culture and the transition to seeing quality as a driver of business success rather than a mere steppingstone to compliance.
In this article, the interview continues with a focus on Sharp’s evolution to a mature Pharmaceutical Quality System (PQS), discussing the five phases involved in transforming the business of quality from foundational compliance to sustained excellence.
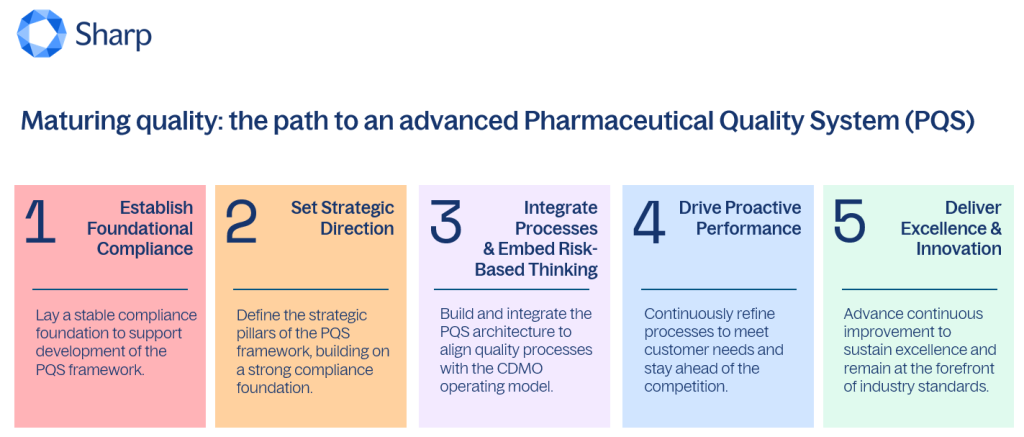
Q: What does it mean for a CDMO to mature its Pharmaceutical Quality System?
Grace Breen (GB): For CDMOs to thrive in an increasingly complex and fast‑changing environment, quality must be enabled by a PQS framework. That PQS must be built to evolve, scale, and remain fit for purpose over time, because quality sits at the intersection of patient safety, supply reliability, and business performance.
I believe the future of quality in CDMOs lies in moving beyond reactive compliance toward a proactive, performance‑driven, and innovation‑enabled quality culture. As a CDMO matures, its PQS framework must mature alongside it, shifting from simply meeting requirements to actively enabling consistent execution, informed decision‑making, and sustainable growth.
At Sharp, this evolution has been intentional. We have guided our journey through a clearly defined five‑phase approach—one that builds deliberately on a strong foundational base and progressively strengthens governance, integration, and performance. This structured evolution has allowed quality to function not just as a safeguard, but as a strategic enabler of our business and a catalyst for long‑term value creation.
Q: What characterizes Phase 1: Foundational Compliance?
GB: Phase 1 is defined by a focus on foundational compliance—establishing core quality processes, training teams on SOPs and cGMP fundamentals, and ensuring that basic data integrity expectations are met. At this stage, quality is typically reactive and viewed as a necessary cost of doing business rather than a contributor to performance or value. While essential for establishing the license to operate, this phase is also an important inflection point—until the basics are reliably in place, an organization cannot move toward proactive risk management, right‑first‑time execution, or a quality culture that truly supports the business. At Sharp, we have been deliberate about recognizing this phase for what it is: a critical foundation that needs to be functional and assure the basics before further maturing the PQS.
Q: How does Phase 2 begin to set strategic direction?
GB: At Sharp, Phase 2 represents the point at which a stable compliance foundation allows us to shift from simply operating the system to intentionally shaping it. This is where the strategic pillars of our PQS begin to take form—translating customer and patient needs into clear requirements, codifying expectations through global quality standards, SOPs, and specifications, and establishing consistent accountability across the organization.
In this phase, leadership commitment becomes visible. Quality is no longer implicit; it is explicitly reflected in enterprise goals, performance metrics, and management expectations. Communication increases, and quality conversations move beyond individual events to focus on direction, priorities, and alignment with the business.
At the same time, we begin to look inward. Cultural assessments help us understand how work is actually being done and whether our behaviors align with the mindset required to support right‑first‑time execution. Risk‑based thinking, grounded in ICH Q9 principles, is introduced across functions to drive prioritization and decision‑making. Management review cadences are established to strengthen governance and create regular, forward‑looking dialogue about system health and performance.
This phase is also where we start to ask a more fundamental question: Is our training enabling people to execute right the first time, or simply confirming procedural knowledge? Phase 2 at Sharp is ultimately about setting direction and intent, creating the clarity, structure, and leadership alignment needed to support the next stage of PQS maturity, where integration and performance take hold.
Q: What distinguishes Phase 3: Integration and risk-based thinking?
GB: In this phase, Sharp’s PQS matures to being fully integrated with our CDMO business model. Processes are integrated across the product lifecycle, interconnected through feed-forward and feedback loops, and designed to be scalable, pragmatic, and fit for purpose as our portfolio and footprint evolve. Quality Risk Management, grounded in ICH Q9 principles, is no longer an activity reserved for specific events; it becomes embedded in everyday decision‑making across functions.
Also in this phase, data-driven decisions are expected and learned intuition is encouraged. Leading indicators, such as process intervention rates and environmental monitoring trends, are deployed. Knowledge management is formalized in line with ICH Q10, and contamination control strategies are established for sterile operations. The focus moves decisively from quality inspection to quality assurance.
This is also the phase where Sharp begins to reframe quality as a value driver, protecting revenue through right-first-time execution, accelerating speed-to-market, and strengthening customer confidence and trust.
Q: What defines a proactive, performance-driven organization in Phase 4?
GB: As quality at Sharp continues to mature, there is a shift from retrospective performance review to predictive, risk‑based decision‑making, supported by data, science, and technology rather than reactive compliance alone. Specifically, phase 4 is characterized by:
- Use of leading indicators and trend analytics that provide early signals of risk, variability, and opportunity across the product lifecycle—using dashboards, and global KPI governance to enable proactive management of quality performance and inspection readiness.
- Use ofProcess Monitoring & Control and capability analysis to proactively support the establishment and maintenance of a state of control, while also enabling continuous improvement through data‑driven identification of adverse trends and improvement opportunities.
- Integration of digital QMS platforms, global dashboards, and evolving analytics capabilities to support near real‑time visibility into quality performance, improving consistency across sites, and strengthening governance and management review effectiveness. Technology is embraced as a competitive advantage to enable smarter, more agile quality decision‑making.
- EmbeddingQuality by Design (QbD) principles into product development and process changes to ensure that quality is built into product and process design rather than inspected in at later stages. A lifecycle-based approach is used to deliver robust product realization, reduce variability, and strengthen knowledge management across the product or service lifecycle.
- Rolling out progressive training and learning strategies to progress from basic compliance training to role‑based learning programs designed to build demonstrated competency, strengthen technical excellence, and reinforce quality culture across the organization. Training effectiveness is increasingly evaluated through performance outcomes, investigation trends, and root cause analyses, rather than completion metrics alone.
- Leadership that is engaged and visible and senior leaders actively participating in management review, inspection readiness, and quality governance, reinforcing accountability, prioritization, and the message that quality is integral to business performance and patient outcomes—not a functional silo.
- A structured approach to proactively anticipating regulatory change and assessing applicability.The PQS must continuously adapt to emerging guidance, technology, and innovation such as the use of AI, to ensure timely, risk‑based implementation across sites.
This phase of maturity sees quality functioning as a strategic business enabler—predictive, integrated, and continuously improving and supporting patient safety, regulatory confidence, and customer trust.
Q: What does sustained excellence look like in Phase 5?
GB: At this stage, quality excellence is defined by Sharp’s ability to sustain, adapt, and lead as industry expectations continue to evolve. The PQS operates as an integrated platform ensuring compliance, and actively enabling innovation, operational excellence, and customer confidence across the enterprise. It is fully embedded into how the business is run, supporting product lifecycle decision‑making, enabling proactive risk management, and maintaining a durable state of control while continuously improving performance. Specifically, phase 5 is characterized by:
- A technology‑enabled PQS with emerging AI applications supporting exception‑based workflows, real‑time monitoring, and predictive insight, simplifying processes while maintaining a robust state of control.
- Knowledge‑driven continuous improvement that enables systematic learning, effective change management, and continuous performance improvement across sites and product lifecycles.
- Quality is owned by leaders at all levels, reinforced through visible engagement, strong governance, and sustained investment in leadership development and capability building.
- Proactive regulatory intelligence and routine maturity assessments, including benchmarking against peers, ensuring Sharp anticipates change and continuously aligns with evolving industry and Quality Management Maturity (QMM) expectations.
Excellence at Sharp is no longer a destination; it is the ability to sustain performance, adapt to change, and continuously improve, fulfilling our responsibility as stewards of the patient promise.
Thank you for reading Part 2 of this Q&A. If you missed Part 1, which covers the overarching and evolving role of quality in a CDMO environment and the leadership strength required to succeed in today’s industry, please click here. Alternatively, learn more about Sharp by downloading the whitepaper below: a step-by-step guide to the commercial packaging process during drug launches.



