
Clinical trial sites and sponsors are turning to eSource to ease pressures over costs and unlock efficiencies, with a recent GlobalData survey revealing how awareness and adoption rates are increasing.
Against an industry backdrop of sponsors and clinical research organizations (CROs) having to do more with less, an electronic solution capable of saving time and money on data collection and management at trial sites is of considerable appeal.
The GlobalData survey highlighted how small and mid-sized biotechs see the potential for eSource to deliver savings, particularly when it comes to CRO vendor costs. There are also time-saving benefits from eSource. This is also why 50% of small and mid-sized biotechs in the survey expect their CROs to embrace this technology and drive implementation and adoption rates. As the clinical landscape becomes increasingly data-driven, a further appeal of eSource is its capacity to deliver greater data accuracy at higher quality to support regulatory submissions.
How eSource is becoming a deciding factor for clinical trial sites
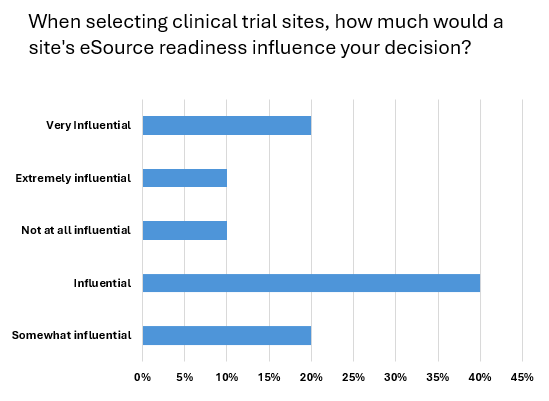
In the survey, 70% of respondents confirmed they were very familiar with the concept of eSource. Alongside this, eSource-readiness is a clear deciding factor for selecting clinical trial sites, with a combined 90% of respondents stating that it was an influence on decision-making. Breaking this down further, 40% said eSource-readiness was influential in selecting clinical trial sites, 20% said it was somewhat influential, while 20% also stated it was very influential. eSource-readiness was extremely influential when deciding trial locations among 10% of respondents. At the other ends, to emphasise the growing importance of eSource, just 10% of respondents said that the tech was not influential.
Conducted across GlobalData’s healthcare network from December 2025 to March 2026, the survey received responses from a total of 161 senior level professionals involved with clinical operations and strategy across biopharma, with 80% from companies with headcounts under 4,999. This engagement suggests there is a higher interest in data management tools at smaller and medium sized biotechs that likely have greater budgetary constraints than larger companies in clinical research. Any solutions that can help to optimize costs and resources, as well as save time, offer major competitive advantages.
This is reflected in the survey responses, where the ability to lower overall trial costs was seen as the main appeal of eSource-enabled sites – with 60% of respondents citing cost-savings as an associated advantage. There was also strong interest in faster database lock/fewer queries and improved data accuracy and quality – both scoring 50% from respondents.
“These results align with the industry’s ‘do more with less’ focus, where digitization is being used to improve speed, quality, and scalability without proportional increases in resourcing,” says Sonnika Lamont, senior pharmaceutical analyst at GlobalData. “The growing use of electronic data capture and eSource signals a shift away from manual, duplicative workflows toward more standardized, efficient clinical trial execution across the pharmaceutical industry.”
How clinical data capture and management are evolving
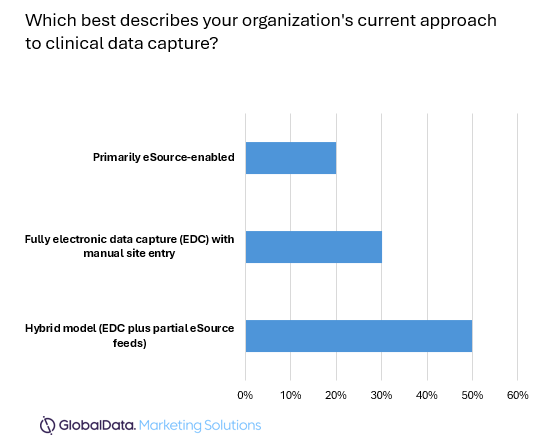
The industry is undergoing an evolution for how clinical data is capture and managed. The majority of clinical data capture uses among respondents was in hybrid models, combining EDC with partial eSource feeds. Notably, just 30% of respondents were still using manual site entry, suggesting that a major shift is underway in the way that clinical data is captured as eSource to EDC adoption rates increase. Initially, eSource to EDC solutions saw a spike in uses during the COVID-19 pandemic, given restrictions on movement when decentralized clinical trials (DCTs) also increased.
In 2023 and 2024, there was a sense that paper-based processes were returning as the industry settled after the pandemic. However, the twin factors of shrinking budgets and increased trial numbers have more recently driven deployment of eSource to EDC solutions out of necessity. Crucially, these changes could be permanent, according to Lamont.
“EDC and eSource are now widely used across the industry, and 2025 marked a new high specifically for clinical trials using EDC, showing that adoption is not only accelerating but becoming embedded as standard practice,” adds Lamont. “While the previous peak occurred in 2021 when COVID-19 drove rapid, necessity-led digitization, 2025’s levels are 22% higher than that pandemic-driven high. That step up beyond an exceptional forced adoption period suggests the shift is durable. eSource to EDC solutions are being adopted for their ongoing operational benefits, including lowering trial cost, reducing site and patient burden, and improving data quality and timeliness.”
Driving eSource implementation in the long-term
Looking ahead at further industry adoption, the fact that 50% of respondents consider CROs to be the overwhelming party responsible for driving eSource adoption is somewhat surprising. Any wider industry roll-out would be expected to be driven by regulators, with this option only attracting 30% of respondents.
However, there is an advantage in CROs taking the lead with eSource as they become familiar with the capabilities ahead of regulators. This would provide greater ownership of data and processes before changes are enforced from the top.
What could drive adoption rates further is the proven benefits of platforms integrating eSource, with more precisely gathered data to support regulatory applications.
The GlobalData survey was sponsored by Paradigm Health, which is a specialist provider of a platform that directly incorporates eSource through comprehensive integrations with EHR platforms, proven to shorten study timelines and reduce costs by strategic uses of automation. The company focuses on increasing access to clinical trial opportunities for more patients, particularly within community and rural settings that are often excluded from trial participation opportunities due to geography, enabling greater democratization of treatment options.
To learn more about the unsustainability of clinical trials, the solutions, and Paradigm Health, download the document below.



