
Saudi Arabia is primed to emerge as the Middle East’s key hub for clinical trials, driven by both sustained state investment and rapid healthcare expansion. As the region’s largest pharmaceutical market, with modern hospitals, widespread clinics, and free or reimbursed access to medicines, the Kingdom already offers a large, well-treated patient base, ideal for diverse and scalable studies.
Saudi Arabia’s healthcare and pharmaceutical markets are currently entering a strong growth phase. According to GlobalData, total healthcare spending is projected to rise by 4.7% in 2026, reaching $61.05m, with per capita expenditure increasing from an estimated $1,692.21 in 2025 to $1,741.75 in 2026. In parallel, pharmaceutical sales in Saudi Arabia are predicted by GlobalData to grow by 6.07% in 2026, climbing to $17.23bn. These increases underscore the growing importance of healthcare and life sciences as strategic investment areas.
A key driver of this growth is the Kingdom’s Health Sector Transformation Programme (Vision 2030[i]), which, underpinned by mandatory insurance, is expanding the public-private ecosystem. The programme’s ambition is for a more comprehensive, effective, and integrated health system that prioritises innovation, financial sustainability, and disease prevention, while also expanding e-health services and digital solutions.
This transformation includes the launch of the SEHA Virtual Hospital[ii], which is connected to more than 150 hospitals to offer approximately 30 specialised services. By supporting virtual consultations, remote diagnostics, and seamless e-prescribing across a national network, SEHA expands access to specialist care for a wider section of the population, simplifies follow-up, and improves data capture.
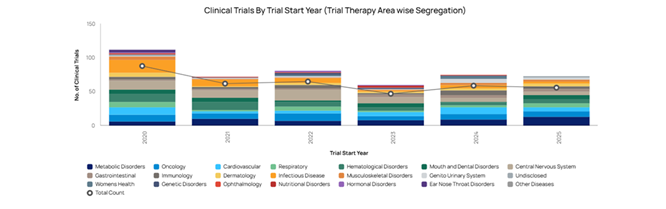
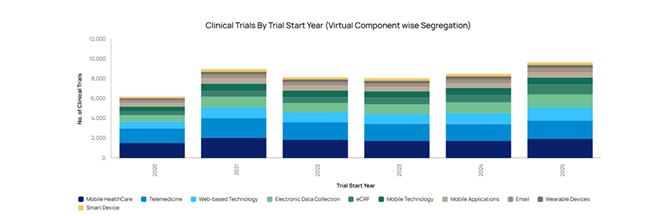
Creating the foundations for future clinical trials
Saudi Arabia’s Vision 2030 is creating the structural foundations for a strong clinical trials environment. By expanding access to care, modernising hospitals and clinics, and encouraging greater private-sector investment, Vision 2030 is producing more research-ready sites with contemporary infrastructure, smoother patient referral pathways, and more stable, well-resourced settings for complex or long-term studies.
The Kingdom’s network of general and specialist hospitals, together with thousands of clinics, polyclinics, and mobile facilities, gives sponsors and contract research organisations (CROs) a wide range of potential trial sites across different regions and care settings. This diversity makes it easier to recruit large, representative study populations and to run multicentre protocols within a single national system.
At the same time, the shift toward greater private-sector participation and the introduction of mandatory health insurance for private-sector workers are deepening the system’s readiness for trials. Compulsory insurance supports sustained demand for healthcare services throughout the year. While growing private investment increases capacity and familiarity with international research practices. The resulting mixed public-private ecosystem expands the variety and capability of potential trial sites, offering more options for sponsors looking to match protocol needs with specific types of facilities.
In addition, rising rates of conditions such as diabetes and cardiovascular disease, combined with strong projected growth in healthcare and pharmaceutical spending, make Saudi Arabia a strategically important location for sponsors targeting high-burden therapeutic areas. This epidemiological profile makes the Vision 2030 infrastructure especially relevant for sponsors focused on chronic and lifestyle diseases who require large, well-characterised patient populations.
The challenges and opportunities for clinical trials in Saudi Arabia
While Saudi Arabia aims to become a regional hub for clinical trials, several barriers are slowing progress. Despite strong infrastructure, specialised expertise is concentrated in major centres, limiting complex studies and diverse recruitment. Alongside this, low awareness, cultural hesitancy, and limited understanding of trial benefits can hinder patient enrolment.
On the logistics side, distributing investigational products and samples across a large, geographically dispersed country increases cost and risk, particularly for maintaining temperature control and compliance in remote sites and warm climates. The evolving public-private healthcare mix adds further complexity that must be navigated, with variable procedures, data standards, and coordination between providers.
Additionally, successfully integrating into the global clinical trials ecosystem demands continuous improvement in data quality, ethics, transparency, and innovation, supported by close collaboration between government, academia, healthcare providers, and international partners.
How Oximio is developing clinical trial capabilities in Saudi Arabia
Logistics providers such as Oximio play a crucial role in ensuring the operational success, safety, and compliance of multicentre clinical trials, and employ several key strategies to mitigate supply chain challenges. Oximio brings more than two decades of global clinical trial logistics experience in applying strict Good Distribution Practice (GDP) standards and proven project management to deliver tailored, end-to-end supply chain solutions. From sourcing and warehousing to temperature-controlled transport, site delivery, and regulatory documentation, Oximio allows sponsors to consolidate these touchpoints with a single specialist provider, reducing operational risk and boosting efficiency.
Oximio has opened its first-of-its-kind facility in Saudi Arabia, with services solely dedicated to clinical trials. A critical element of this approach is investment in advanced storage and transport infrastructure, including state-of-the-art, GDP-compliant warehouses capable of handling medicines and ancillary supplies under precisely monitored ambient and cold-storage conditions.
Temperature monitoring throughout storage and transit ensure that products remain within prescribed limits, vital for the integrity and validity of investigational drugs and samples. As deviations can compromise both the scientific quality of a study and regulatory compliance, these controls are essential in regions such as Saudi Arabia, where climate extremes present additional risk.
By providing a single point of coordination for imports, customs clearance, destruction of expired stock, and return logistics, Oximio mitigates regulatory risk and reduces administrative burdens for sponsors unfamiliar with local requirements. This integrated approach reduces handovers and the risk of miscommunication, critical for ensuring that trials progress smoothly with minimal loss or delay.
Saudi Arabia: A growing regional hub for clinical research
Advanced clinical logistics capabilities, provided by specialists such as Oximio, ensure compliant, temperature-controlled movement of drugs and samples, enabling complex multicentre trials. Together, these strengths mean Saudi Arabia isn’t just a growing market – it’s becoming a regional hub for clinical research and a key driver of innovation and better health across the Middle East.
To learn more about clinical trial locations in the Middle East, download the document below.
[i] https://www.vision2030.gov.sa/en/explore/programs/health-sector-transformation-program
[ii] https://oecd-opsi.org/wp-content/uploads/2022/10/Overview-SVH-2.pdf



