
As clinical trial protocols grow more complex, so does the burden on research sites managing data collection and entry. Manual data entry (already time-consuming and error-prone) has tripled over the past decade, further straining site resources
and delaying access to critical insights. On average, it takes eight days from a patient visit to data entry, slowing trial progress and inflating operational costs.
While eSource has the potential to ease this burden, siloed systems prevent both sponsors and sites from realizing its benefits. To improve efficiency and data quality, the industry needs purpose-built solutions that streamline data collection, reduce site workload, and enhance collaboration—without adding operational overhead or delaying trial timelines.
Reduce data capture burden to boost efficiency and research capacity
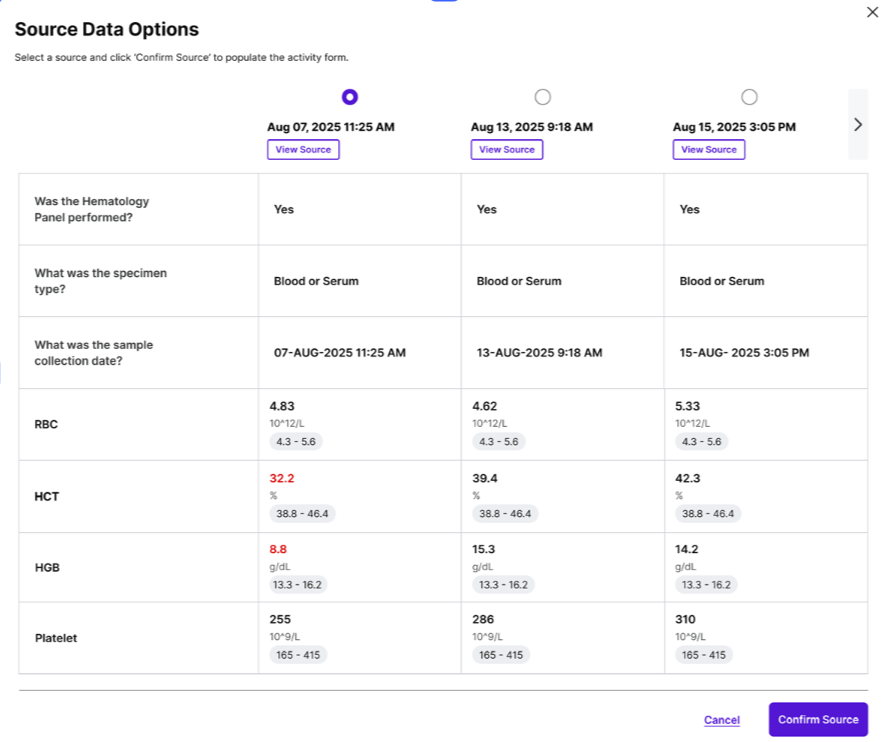
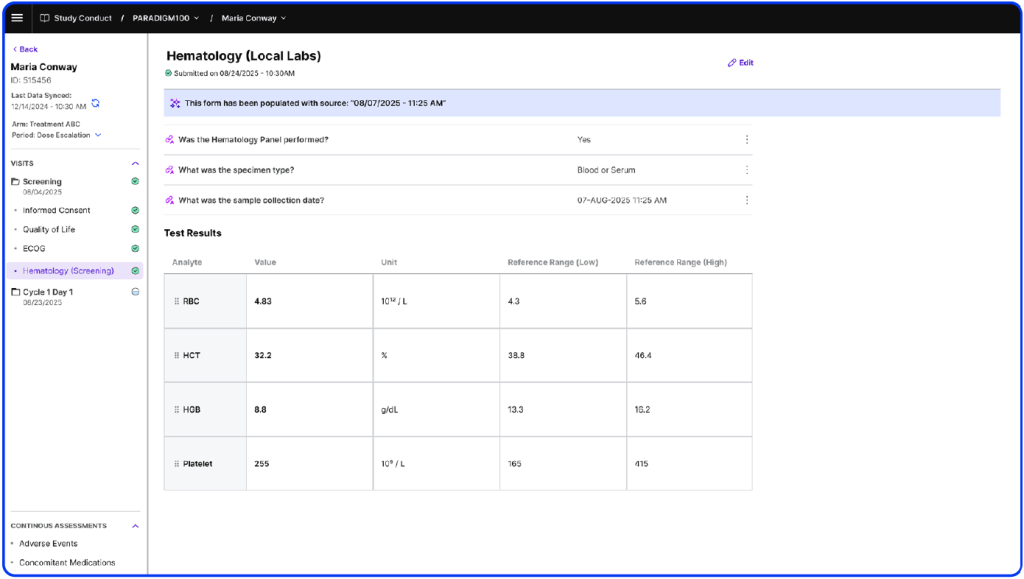
eSource Casebook is an EHR-integrated application that extracts trial-relevant data from structured and unstructured sources, auto-populates eCRFs, and transmits data to sponsor EDCs. By taking over the site-facing portion of the sponsor EDC and pulling in all source data, eSource Casebook consolidates data entry into a single application, seamlessly integrating into research team workflows.
Clinical Research Coordinators and research teams can enter data faster using an intuitive import workflow that includes citations for quick verification. By saving significant time and reducing data burden, sites can enhance the research experience, take on more studies with fewer resources, and ensure a smoother trial process for patients
Efficient and accurate data management
- Single interface for data extraction, verification, and eCRF population to reduce manual effort
- AI-powered tools accelerate data collection and minimize errors, supporting cleaner and faster submissions
- Direct Data Capture and paper source upload support ensures complete data capture regardless of source format
Empowered and productive research teams
- Customizable workflows and intuitive design reduce training time and accelerate implementation across sites
- Access to structured and unstructured EHR data enables comprehensive, real-time data collection from within existing research workflows
- Integrated molecular diagnostics data feeds simplify the inclusion of genomic and lab data
Scalable and dynamic infrastructure
- Consolidates tools by replacing the site-facing EDC, minimizing clicks and context-switching
- Direct integration with EDCs and source systems for realtime, standards-based data flow with minimal configuration
- Flexible architecture supports growth across trials and therapeutic areas
Connect to the EHR and integrate into existing workflows
Directly import structured EHR data via FHIR and leverage LLMs to extract detail from unstructured data to minimize manual entry.
Ready to accelerate data entry, reduce auditing costs, and ensure faster, more accurate data flow?
Contact Paradigm Health at paradigm.inc/solutions/conduct-esource-casebook.



