Cell therapies have gained steam in oncology since Novartis’s Kymriah (tisagenlecleucel) became the first chimeric antigen receptor (CAR)-T cell therapy approved by the FDA. There are currently 12 cell therapies marketed in the US out of 25 approved worldwide. The majority of marketed cell therapies are autologous, requiring patients to undergo leukapheresis to donate their own cells, which are subsequently modified ex vivo and reintroduced into the patient. There are several significant disadvantages to requiring the patients’ cells in the cell therapy manufacturing process.
Patients who are eligible for cell therapies are typically heavily pre-treated with chemotherapies and have haematological malignancies, which may compromise the quality of the initial blood collection, reducing the likelihood of the cell therapy manufacturing process being successful. The patient will also have to travel to a qualified leukapheresis centre, and these are typically located in urban areas and can therefore involve significant travel costs for the patient. The patient will also need to wait between leukapheresis and administration of the cell therapy. This vein-to-vein time, which can take weeks, will often require a bridging therapy to be administered, which can trigger serious adverse events.
Figure 1: Proportion of autologous versus allogeneic cell therapies in oncology by stage of development
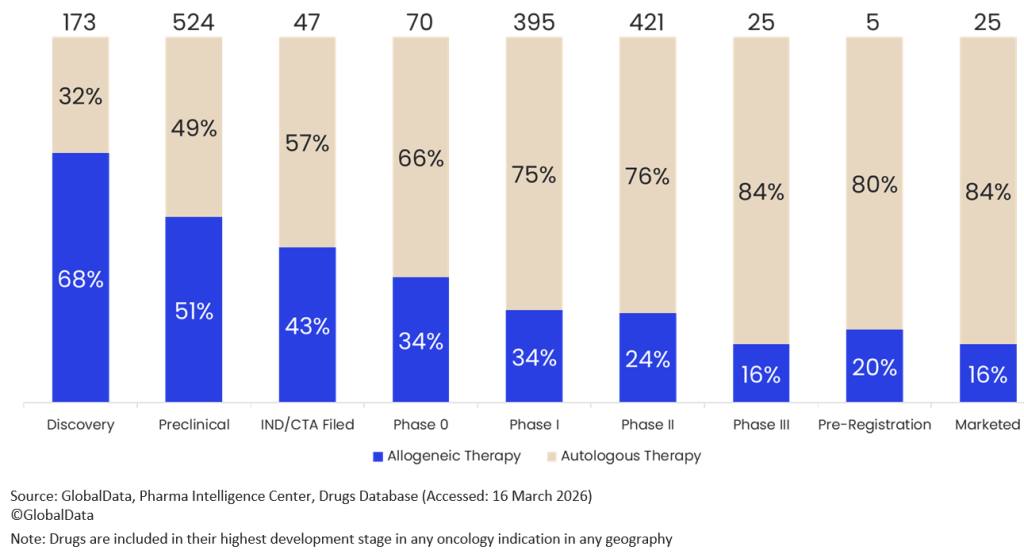
These factors reduce the uptake of existing autologous CAR-T therapies. One strategy that would counteract these disadvantages is to develop off-the-shelf allogeneic cell therapies. These therapies typically depend on donors whose cells can be transfused into many patients. Pharmaceutical companies are shifting from the development of autologous cell therapies to allogeneic cell therapies: 68% and 51% of cell therapies in discovery or preclinical development for oncology indications are allogeneic, compared to just 24% and 16% of those in Phase II or III clinical trials. This increase in earlier stages of development is indicative of an increase in desire to meet the challenges of developing allogeneic cell therapies, of which there are many.
Allogeneic therapies come with a significant risk of inducing graft-versus-host disease (GvHD), which is counteracted by knocking down the expression of the T-cell receptor (TCR) of the donor’s cells. However, the patient’s natural killer (NK) cells recognise the donor T cells as foreign, mediating their destruction, which reduces the persistence of therapeutic cells and reduces overall efficacy. The extra genetic complexity results in a more significant regulatory complexity, as each extra mutation must be validated for off-target effects, increasing the time needed to produce IND-enabling research. This explains some of the higher attrition rates for autologous therapies before they reach clinical development. There are many companies that remain optimistic that their therapies can overcome GvHD as well as host rejection and develop a persistent, effective allogeneic cell therapy; however, it will be many years before they overtake autologous therapies in the clinic.

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalData




