Innovative pharmaceutical trade associations across the Nordic region have reacted with vigilance to a recent report by GlobalData that highlighted the potential effect on product launch and withdrawal trends in Europe caused by the Most Favored Nation (MFN) policy in the US.
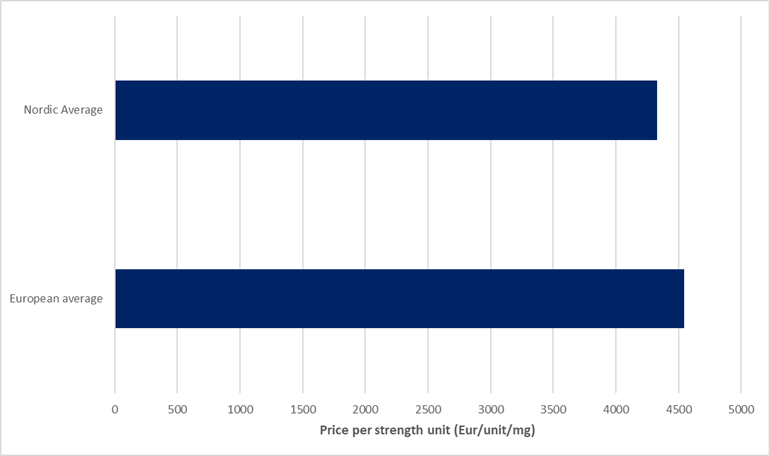
Recent GlobalData Price Intelligence (POLI) analysis showed a 35% decline in pharmaceutical launches across Europe in the ten months after the introduction of international reference pricing (IRP) in the US. While other factors such as regulatory delays, pricing and reimbursement negotiations, supply chain constraints, manufacturing constraints, or failed site inspections, in addition to MFN, will be at play, impacting the decline in launches, it has nonetheless drawn close attention from the Danish Association of the Pharmaceutical Industry (LIF) and Swedish Association of the Pharmaceutical Industry (LIF), respectively. A report by Sweden’s LIF spotlights POLI data to show that the first effects of MFN on patient access to new medicines in Europe can already be detected. Indeed, POLI suggests that Sweden has been disproportionately impacted by product launch delays and product withdrawals since the imposition of MFN. This is linked to ex-manufacturer prices in the Nordic countries being below the European average. Based on GlobalData Price Intelligence analysis of a sample of 21 oncology products that received regulatory approval after 2020, and that are priced in 20 or more European markets, the average price per strength unit (€/unit/mg) is 5% lower in the Nordic markets combined compared to the European average.
Figure 1: Average price per strength unit of selected oncology products distributed by the European and Nordic average
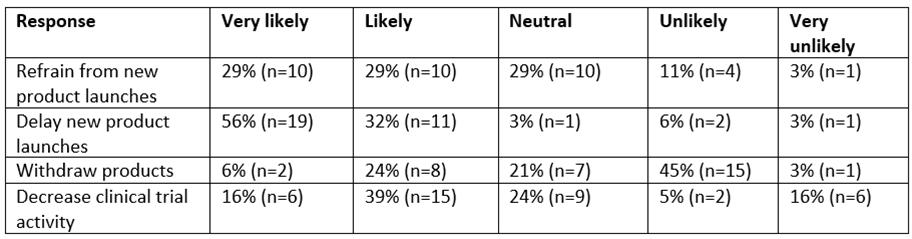
The prevailing concern that MFN could damage access to medicines in the Nordic region is also reflected in an industry survey of pharmaceutical companies carried out by Sweden’s LIF trade association, the results of which show a large majority of industry respondents anticipate major shifts in launch strategies in the region over the coming years. The LIF industry questionnaire compares to a GlobalData State of the Biopharmaceutical Industry survey, published in December 2025, in which respondents were asked whether the US MFN policy will or will not impact their company’s strategy. At the time, GlobalData survey respondents were split 41% “yes” to 32% “no” with 27% undecided. The LIF survey suggests that “yes” is now increasingly the default answer for most pharmaceutical companies operating in Sweden and the wider Nordic region.
Table 1: LIF industry survey of MFN implications for the Swedish market in the next 0–3-year outlook
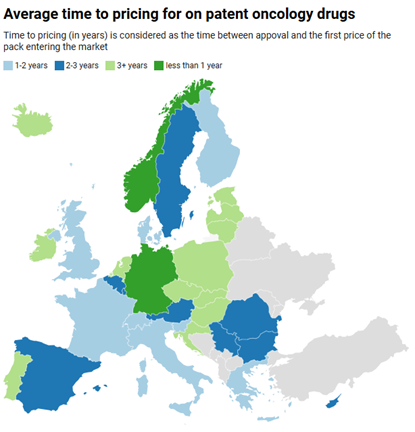
Denmark’s LIF trade association also shows concerns over these early signals. Its reaction warns that the threat posed by US pricing policies, combined with low list prices in Denmark, presents a serious risk that the average time to launch will slip. The graph below represents the current average time to pricing (in years) for on-patent oncology products across the European region.
Figure 2: Average time to pricing for on-patent oncology products distributed across the European region
Industry stakeholders in Nordic countries are not alone in paying close attention to MFN developments. Policymakers are mobilising government resources to investigate the potential impacts and potentially propose reforms to mitigate negative effects. For instance, Denmark’s Ministry of Health and the Ministry of Industry, Business and Financial Affairs recently established a task force to investigate the effects of MFN policy, while Sweden’s TLV agency has been commissioned to prepare a report examining the risks MFN poses to the sustainability of the pricing system. A preliminary report by the TLV was somewhat underwhelming, with the analysis confined to noting the “many uncertainties” in how US policy changes could affect medicine prices, launch strategies, and access conditions, before concluding that it is still “too early to say” whether targeted measures by Sweden are needed and, if they are, what these targeted policy measures should be. It can only be hoped that a fuller analysis, coupled with reform proposals, will emerge when the TLV delivers a final report in late 2026. In the meantime, industry trade associations already see the writing on the wall and are collectively appealing for national pricing and reimbursement (P&R) system overhauls. This sense of urgency is perhaps seen in the proposals emanating from the opposition Progress Party (FRP) in Norway, which recently called for the implementation of inflation-adjusted rises in willingness-to-pay (WTP) thresholds for cost-effectiveness, a proposal that is slowly gaining momentum since GlobalData reported on initiatives in this area in late 2025, and also has overwhelming support from the innovative sector. The head of the LIF in Denmark, Thomas Senderovitz, is also reported to have said that frameworks for pharmaceutical spending needed to be raised, and that reform of cost-effectiveness thresholds should be prioritised. Planned amendments to new medicine evaluation criteria and price ceiling regulations in Sweden will also potentially be finalised in 2026.