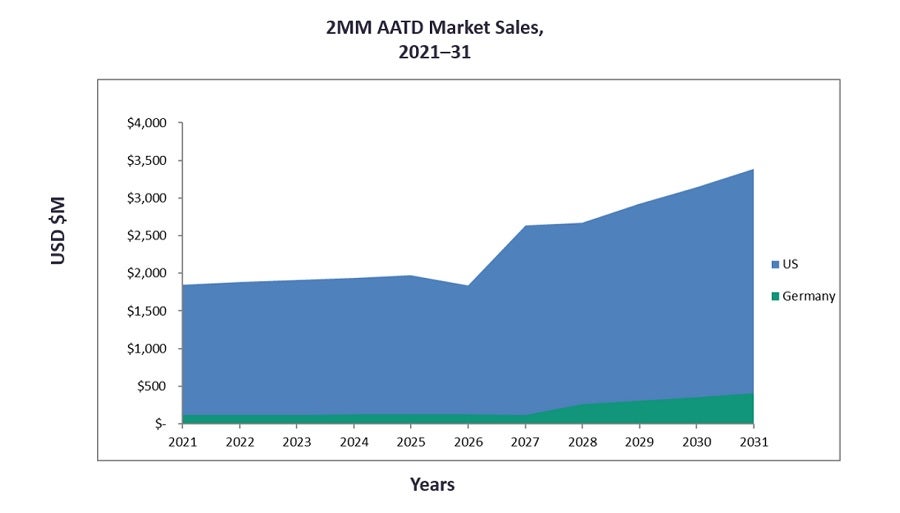
According to GlobalData’s recent Alpha-1 Antitrypsin Deficiency (AATD): Opportunity Analysis and Forecast to 2031 report, the AATD market is expected to see significant growth from 2021 to 2031. The two major markets (2MM: the US and Germany) will increase in market size from $1.20bn last year to $3.48bn in 2031, at a compound annual growth rate (CAGR) of 11.2%. This sales growth will be in line with a steadily increasing disease prevalence and the entrance of novel agents into the market. Several drugs in the pipeline have novel mechanisms of action (MOAs), including novel neutrophil elastase (NE) inhibitors, alvelestat and PHP-303, and two novel RNAi therapies, belcesiran and ARO-AAT.
AATD is a rare inherited disease resulting from the deficiency in the production of alpha-1 antitrypsin (AAT) protein due to mutations in the SERPINA 1 gene. Low levels of AAT result in damage to lung tissue, leading to chronic obstructive pulmonary disorder (COPD), while mutated AAT protein can also accumulate in hepatocytes, leading to cirrhosis. The current treatment for AATD involves the use of augmentation therapy, alpha-1 proteinase inhibitors (AI-PIs), whereby patients are administered intravenous AAT, allowing patients to have an increase in serum AAT and help to reduce the decline in lung function that occurs in COPD.
AI-PIs are the only marketed therapies currently in this space, and they are only able to directly treat AATD-related lung disease, but not the liver disease that results from the accumulation of misfolded AAT. There are four AI-PIs available across the 2MM, with the US having all four marketed products and Germany having two marketed therapies: Takeda and Kamada’s Glassia (US only), Grifols’s Prolastin-C (US and Germany), CSL Behring’s Zemaira (US)/Respreeza (Germany), and Baxaltas’s Aralast NP (US). The patents for these AI-PIs are expired and the AATD market remains in need of therapies with novel MOAs that are able to either reverse the decline of lung function or prevent the production of mutated AAT that leads to cirrhosis.
There are currently several therapies in Phase II of clinical development, three of which are targeted to AATD-related emphysema and two other therapies targeted at liver disease. Although this will provide more efficacious treatment options for AATD patients, an unmet need remains for therapies that prevent the decline in lung function, mainly by reversing the decline in forced expiratory volume (FEV1) and/or liver fibrosis, and significantly improve long-term outcomes.
Therapies with improved efficacy and reduced cost all remain largely unmet needs, as AI-PIs are often unable to prevent the long-term damage that occurs in COPD patients and may only slow down disease progression. In addition, improvements in early diagnosis of AATD are critical to preventing some of the irreversible damage that occurs in both lung and liver disease patients, with most adults presenting with irreversible emphysema and some liver function decline and paediatric patients presenting with liver failure. There are also other drawbacks to the current AI-PI regimen, such as treatment dosage being poorly standardised, with a fixed dosage of 60mg/kg with a protective AAT threshold of 11µm proving to be an inaccurate measure of dosage and monitoring, as the half-life of the AAT-protein is four to five days.
Two novel NE inhibitors, Mereo Biopharma’s alvelestat (currently in Phase II in the US and Germany) and pH Pharma’s PHP-303 (currently in Phase II in Germany), have novel MOAs that are likely to reverse the decline in elastin in the lung tissue from overactivity in NE due to lack of AAT that often regulates NE activity. Across the forecast period, it can be seen that AI-PIs will remain a likely first-line therapy for those with AATD lung disease, but key opinion leaders (KOLs) interviewed by GlobalData have reported that NE inhibitors are likely to be used in combination, or as an adjunct therapy, with AI-PI. In addition, although Kamada Pharmaceuticals is developing an inhalable AI-PI (second-generation), KOLs reported that the efficacy of this therapy remains to be determined and are sceptical as to whether adequate AAT serum levels can be achieved.
There are a further two late-stage pipeline therapies, currently at Phase II across the 2MM, that are targeted to treat AATD liver disease directly. AATD liver disease leads to cirrhosis and liver fibrosis due to the accumulation of mutated AAT protein in the hepatocytes. The two therapies, Novo Nordisk’s belcesiran and Arrowhead’s ARO-AAT, are RNAi therapies that are both administered subcutaneously, on a weekly basis, and prevent the production of mutant AAT proteins that lead to long-term fibrosis. These therapies address a real unmet need in the AATD market, therapies that directly target the liver as current AI-PI is unable to do so. Despite this, clinical data demonstrating long-term and impactful reversal of liver fibrosis remains to be seen.
The graph below represents forecast sales for the AATD market.