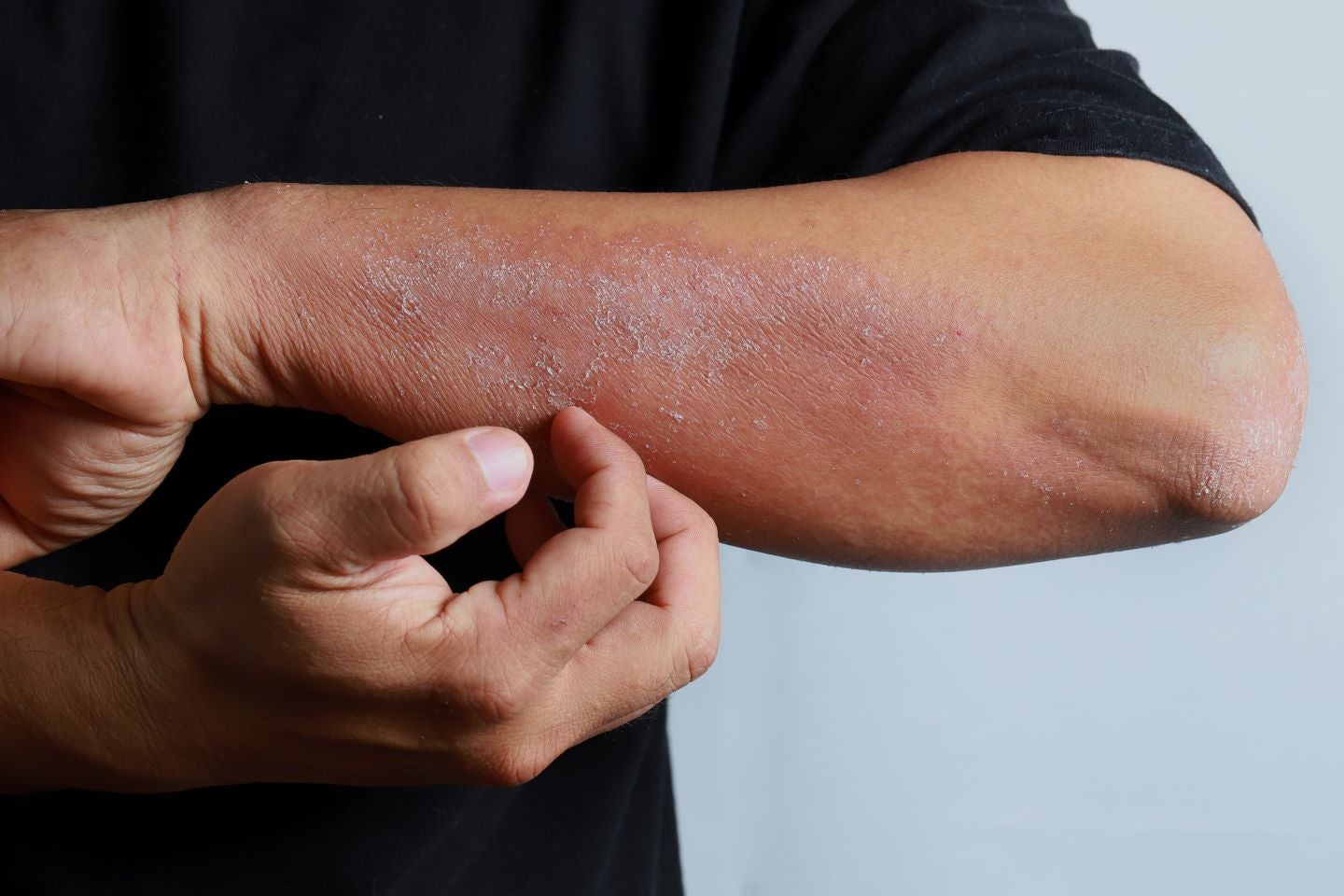
Atopic dermatitis (AD) manifests itself differently across White, Brown, Asian, and Black skin and this impacts the process used for diagnosing these patients. One key study conducted by the Icahn School of Medicine found that in the US, filaggrin (FLG) mutations were one-sixth as common in African Americans (AA) than in European Americans (EA), (Brunner, P. M. and Guttman-Yassky, E. (2019) ‘Racial differences in atopic dermatitis’, Annals of Allergy, Asthma and Immunology). Moreover, Asian patients have a stronger T-helper 17 (Th17) and T-helper (Th22) activation than both AA patients and White patients with AD. It has also been noted that AA patients have darker skin that is more difficult to distinguish clinically from non-lesional skin. Despite the recent advances in both topical and oral dermatologic therapies, it remains difficult to treat the disease, especially in patients with skin of colour.
Targeting and treating patients with skin of colour is a spotlight theme at the 25th World Congress of Dermatology (WCD) conference as exemplified by the various presentation titles selected for the symposia including – AD in Skin of Color- Clinical Presentations and Challenges in Therapy (SY02C), Does diagnosing AD in skin of color need a different criteria (SY36A), Racial/Ethnic Variations in Skin Barrier and the Relationship of Skin Barrier Disruption (LS14), and Ethnic Variations and Cultural Considerations when Targeting African, Asian, and Latin American Patients (LS14). Moreover, Galderma will be holding a symposium titled, Diversity and Inclusion in Dermatology: a Worldwide Profiling of Sensitive Skin (LS10). Through the presentation, Galderma will likely raise the need for a broader representation of patient demographics, focusing on accurate diagnosis and treatment of individuals with diverse skin types.
Over the years, therapeutic options for AD patients have expanded from steroids and emollients to more targeted treatments such as Sanofi and Regeneron’s biologic Dupixent (dupilumab) and Janus kinase (JAK) inhibitors including AbbVie’s Rinvoq (upadacitinib) and Pfizer’s Cibinqo (abrocitinib). The latter therapies are used more for the treatment of moderate to severe AD as they are effective in improving symptoms and quality of life (QoL), but AA patients may require earlier, more aggressive treatment due to their tendency for greater disease severity. Another distinction when treating skin of colour is that patients suffer from lasting hyperpigmentation, more commonly known as post-inflammatory hyperpigmentation (PIH), so understandably doctors would need to diagnose and manage the scarring incurred and this could also impact the patient’s long-term QoL. Therefore, the role of diversity in trial recruitment is becoming more important as the scale of diversity in clinical trials is directly proportional to the results becoming more meaningful for treatment in the real world. To ensure representativeness, clinical trials should enrol people in similar percentages to their overall representation in the population.
The spotlight on the skin of colour will not dim and the theme will continue through many conferences including the WCD 2023. As with the explosion of newly approved therapeutics and increasing clinical activity in AD, it is imperative to understand the clinical, genetic, and molecular aspects of AD across various ethnic groups.