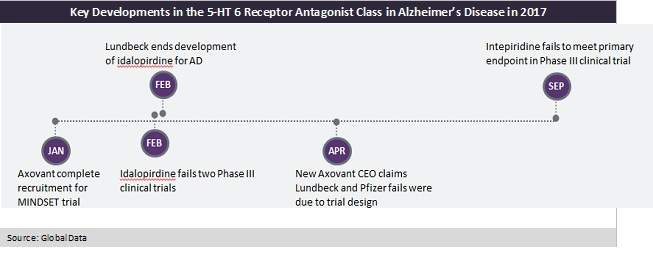
The hit list of failed Alzheimer’s drugs continued to grow and 2017 saw another two names added to the list of drugs that failed to hit the mark in Phase III clinical trials.
Both Axovant’s intepiridine and Lundbeck’s idalopirdine failed their Phase III clinical trials, leaving the Alzheimer’s pipeline no closer to a FDA-approved breakthrough.
After failing its second and third Phase III, in February 2017 Lundbeck terminated the clinical development of its pipeline drug due to weak efficacy. Subsequently, in September 2017, intepiridine was also terminated as it was unable to significantly improve cognition or function for patients relative to placebo. While these results were disappointing they were not surprising, with both drugs failing previous clinical trials and with drugs from the same class failing in previous years; most notably, Pfizer’s PF-0521237.
While some may point the finger at the mechanism of action, scientific theory backs the rationale of blocking 5-HT6 receptors in the brain. The true problem lies in the clinical trial setup, as manufacturers face a balancing act of several factors that include selecting effective dosing to minimize participant drop-out, enrollment of treatment-responsive patients, and lack of robust measurement tools or biomarkers.
The Holy Grail for Alzheimer’s, a disease modifying agent, remains elusive, with the 5-HT6 receptor antagonist class failing to demonstrate efficacy despite a solid scientific rationale behind it. The failure of intepiridine and idalopirdine means there are no more 5-HT6 receptor antagonists in the pipeline which ramps up pressure on other classes, such as the BACE inhibitors and monoclonal antibodies, to deliver a the first next FDA approved Alzheimer’s therapy.