Scale Up and cGMP Manufacturing
You have taken your drug candidate as far as you can. It is time to scale up your process from the bench to clinical or commercial size batches. But is your process robust enough?

Therapure Biopharma is a contract development and manufacturing organization (CDMO) that specializes in the development, scale up and manufacture of complex biologics.
You have successfully submitted your enquiry. Someone from our company will respond ASAP
Therapure Biopharma is a contract development and manufacturing organization (CDMO) that specializes in the development, scale up and manufacture of complex biologics. Therapure operates a modern 130,000ft² cGMP facility in the Greater Toronto area that is equipped with Class A to C clean rooms, QC and analytical labs, clinical and commercial-scale manufacturing suites, mammalian cell culture production capability with upstream and downstream processing, and aseptic fill/finish capacity.
As a specialist in biologics contract manufacturing, Therapure understands your process scale up and cGMP manufacturing needs. We work as an extension of your organization, adding value as your trusted biologics manufacturing partner. Therapure has over 20 years of experience manufacturing a variety of sources of API, including:
Therapure is highly experienced in protein scale up and cGMP manufacturing. When batch manufacturing starts, we encourage clients to be on-site, observing the manufacturing runs with us. Upon completion, we review the run with the client, going through every technical aspect of the batch records in order to fine tune the process for the next run.
Our protein manufacturing capabilities include:
When you are searching for a company to formulate, fill and finish your bulk drug substance into your desired final product container, Therapure is your trusted partner for cost-effective clinical and commercial-scale fill/finish services. Therapure has experience filling and finishing both therapeutic proteins and small molecules into glass vials, preformed IV bags and prefilled syringes.
For glass vial filling Therapure uses Bosch TL equipment and can fill 3,000 x 2ml vials per hour. Container volumes can range from 2ml to 200ml.
Therapure offers integrated lyophilization capabilities at both the clinical and commercial scale.
Clinical-scale lyophilization:
Commercial-scale lyophilization:
Therapure operates a world-class 130,000ft² facility with over 28,000ft² of cGMP manufacturing space. Our cGMP contract manufacturing services are designed to help you every step of the way – from technology transfer and process development, to analytical development and testing, to the scale up and cGMP manufacturing phases and then on to aseptic fill/finish and lyophilization and, finally, a range of support services, including GMP-compliant warehousing, distribution services, validation support, and stability testing services.

You have taken your drug candidate as far as you can. It is time to scale up your process from the bench to clinical or commercial size batches. But is your process robust enough?

More than 60 years old, the East Syracuse campus was launched in 1943.

Switzerland-based biopharmaceuticals supplier Lonza and French life sciences firm Sanofi have established a joint venture (JV) to set up a large-scale biologics production facility in Visp, Switzerland.
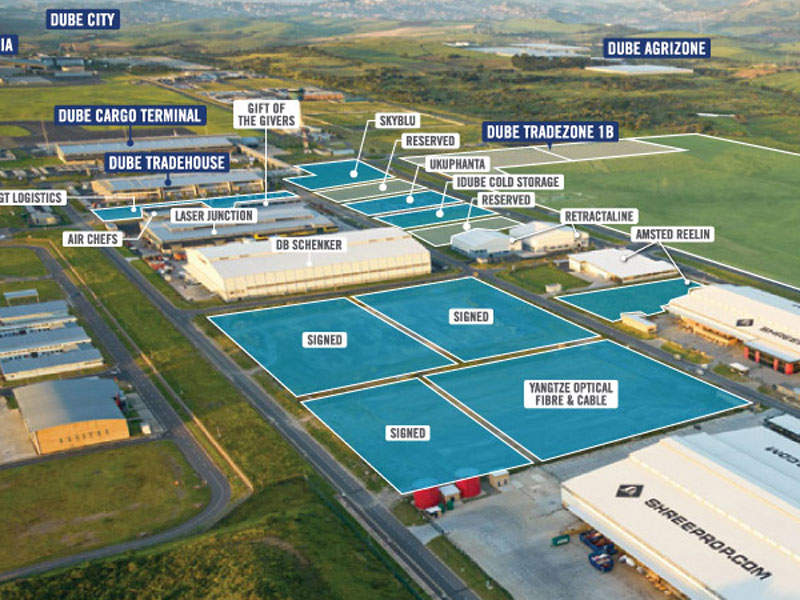
India-based pharmaceutical company Cipla has announced plans to build a manufacturing facility in Durban, KwaZulu-Natal, South Africa.

US-based biopharmaceutical company Baxalta, a spin-off of Baxter International, began building a biologics facility in Georgia, US, in August 2012.

Therapure Biopharma will be attending Bio International Show from 15-18 June 2015.

Therapure Biomanufacturing will attend Interphex at the Javits Center in New York.

Therapure Biopharma is exhibiting at the upcoming Outsourced Pharma West conference and exhibition.

Therapure Biopharma is appearing at the upcoming BioProcess International Conference & Exhibition.

Therapure Biopharma is to host a booth at INTERPHEX in the US from 18-20 March.

Therapure Biopharma, a contract development and manufacturing organization (CDMO) of biotherapeutics, is pleased to announce that the company will participate as a subcontractor for DynPort Vaccine Company (DVC), a CSC company, that was awarded a US cost-plus-fixed-fee contract with a maximum value of $157.3m (prime contract number W911QY-13-C-0056) by the US Department of Defense (DoD) to support the development of a prophylactic countermeasure to prevent the effects of organophosphorus nerve agent poisoning.

Therapure Biopharma is pleased to announce today that it has been awarded the Gold Leaf Award for Emerging Company of the Year by BIOTECanada in the Health Biotech Category for 2012.

Therapure Biopharma has released a white paper entitled 'Scale Up and cGMP Manufacturing'.

Therapure Biopharma, a contract development and manufacturing organization specializing in complex biologics, will be exhibiting at INTERPHEX 2013, New York.

Therapure Biopharma has announced today that its biomanufacturing division, Therapure Biomanufacturing, has won the 2013 CMO Leadership Award as a leader in all five categories: Quality, Reliability, Innovation, Productivity and Regulatory.

Therapure Biopharma Inc has been awarded the Gold Leaf Award for Emerging Company of the Year by BIOTECanada in the Health Biotech Category for 2012.

Therapure Biopharma has officially opened its new custom biologics manufacturing wing, which supports the development and manufacture of innovative medical treatments.

Therapure Biopharma Inc's Custom Biologics Manufacturing Wing officially opened today. The new facility supports the development and manufacture of innovative medical treatments.

Therapure Biopharma has announced that it has received a compliant rating from the Health Products and Food Branch Inspectorate of Health Canada.