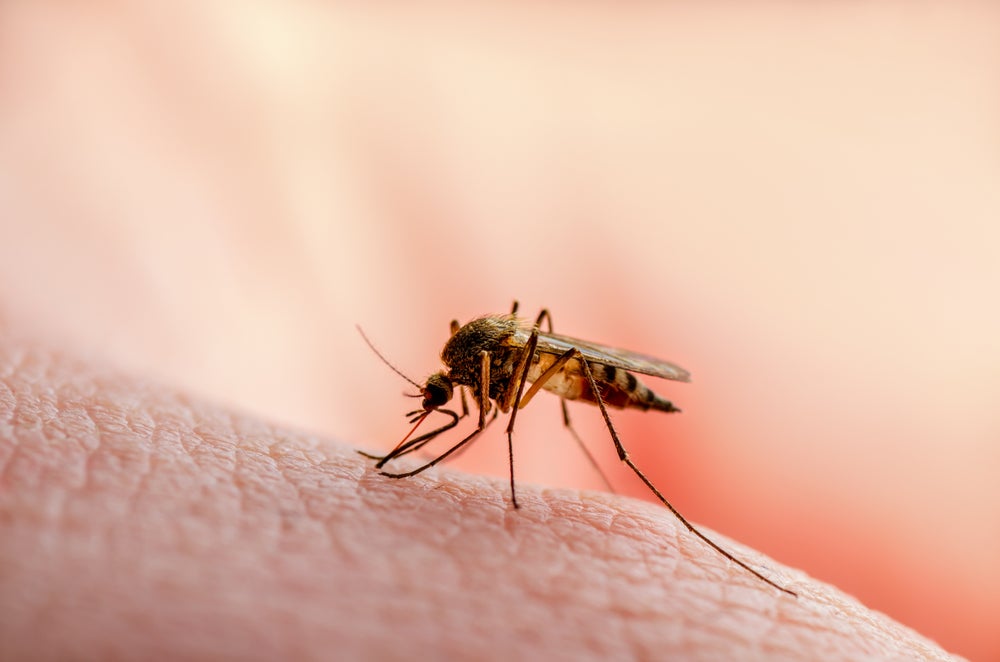
Malaria is a life-threatening disease transmitted to people through the bites of infected female Anopheles mosquitos. Plasmodium parasites are transmitted through the bites, causing initial flulike symptoms that can swiftly progress to more severe symptoms such as kidney failure, seizures, mental confusion, coma and even death if not properly treated.
The disease kills more than 400,000 people a year, mostly children in sub-Saharan Africa. Numerous reasons exist for its prevalence in this region, a key factor being that the mosquito species living in this territory tend to have longer lifespans, giving the parasite time more time to complete its development.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The parasite initially settles into the human liver and then spreads into the blood where it grows and divides. It can hide from the immune system and multiply within human blood cells, binding to a molecule in the blood cell called basigin using a molecule called RH5. Antibodies can stop RH5 from binding to basigin, meaning the parasite is left outside the cell and destroyed, so vaccine candidates for malaria often seek to prepare the body to develop this antibody response.
RTS,S/AS01 (RTS,S), known by the brand name of Mosquirix, is the only approved vaccine for malaria. Developed by the PATH Malaria Vaccine Initiative (MVI) and GlaxoSmithKline (GSK) with support from the Bill and Melinda Gates Foundation, RTS,S is the only candidate thus far to demonstrate a sufficient reduction malaria cases, particularly life-threatening and severe ones, in young African children.
It acts against P. falciparum, the deadliest malaria parasite globally and the most prevalent in Africa. However, its efficacy is low – it prevents only around four in ten cases of malaria – and it needs to be administered in four separate doses.
Researchers at the University of Oxford have now developed a new malaria vaccine candidate, which could prove far more efficacious than RTS,S. Early trials have shown that the jab is 77% effective, and a major study is now in progress.
Medical Technology takes a look back at the history of malaria vaccine development.
1880 – 1897: Mosquitos identified as the vector for malaria spread
The term malaria originates from the mediaeval Italian mala aria which means ‘bad air’, as the disease was formerly believed to be caused by contaminated air. Scientific studies on malaria first began to advance in 1880, when a French army doctor named Charles Louis Alphonse Laveran observed parasites in the blood of infected people and proposed that these were the cause of disease.
A year later Carlos Finlay, a Cuban doctor treating people with yellow fever in Havana, provided strong evidence that mosquitos were transmitting disease to and from humans.
Come 1894, Scottish physicians Sir Ronald Ross and Sir Patrick Manson began to work together. Four years of collaboration culminated in 1897 when Ross was able to prove the complete lifecycle of the malaria parasite in mosquitos.
Ross was able to establish that the mosquito was the vehicle that spread malaria to humans by isolating malaria parasites from the salivary glands of mosquitos that fed on infected birds.
1967: Mice first immunised against malaria
Austrian-Brazilian researcher Ruth Sonntag Nussenzweig demonstrated that mice could acquire immunity to the P. berghei parasite by exposing them to P. berghei sporozoites that had been inactivated by X-ray irradiation.
This provided the mice with significant protection against the parasite when they were subsequently injected with a live version.
2002: Nussenzweig’s research is adapted to humans
A team of researchers led by Stephen Hoffman immunised 11 volunteers against P. falciparum, through the bites of 1001 – 2927 irradiated mosquitos harbouring infectious sporozoites. The radiation meant that, while the parasites could still enter liver cells, they couldn’t divide or infect red blood cells. Over 42 weeks, 24 out of 26 tests on the volunteers showed they were protected from malaria.
2005: RTS,S deployed in Mozambique
RTS,S was used in a randomised controlled trial for the prevention of P. falciparum among 2022 children in Mozambique children aged between one and four. The results were able to confer partial protection among the children for at least 18 months.
RTS,S is a pre-erythrocytic vaccine, which means it targets the infectious phase of malaria and aims to either prevent sporozoites from getting into the cells of the liver in the first place or destroy infected liver cells.
It uses the P. falciparum circumsporozoite protein (CSP) from the pre-erythrocytic stage, fused with the surface antigen from hepatitis B plus a chemical adjuvant to boost the immune response.
The most significant challenge for a pre-erythrocytic vaccine is time, as sporozoites reach the liver following a mosquito bite in under an hour, meaning the immune system has only a short window of time to eliminate the parasite.
2009 – 2014: Phase III trial of RTS,S demonstrates modest protection
The Phase III trial of RTS,S began in May 2009. The study enrolled 15,460 children in seven countries across sub-Saharan Africa, aged either five to 17 months at first immunisation or six to 12 weeks at first immunisation.
Participants were randomly assigned to receive three doses of the vaccine at months zero, one, two and a booster dose at month 20; the three initial doses of RTS,S/AS01 and a dose of a comparator vaccine at month 20; or a comparator vaccine at all stages.
Data from the trial showed that the vaccine-induced modest protection against P. falciparum malaria in children in the 12 months following vaccination, with its efficacy decreasing thereafter.
Among children aged five to 17 months who received four doses of RTS,S, the vaccine prevented approximately 39% of cases of malaria over a four-year follow-up period. It was also able to prevent 29% of cases of severe malaria. The vaccine also reduced the need for blood transfusions, which are required to treat life-threatening malaria anaemia, by 29%.
2015 – 2016: RTS,S receives official approval
In July 2015 the European Medicines Agency (EMA) adopted a positive scientific opinion for RTS,S under Article 58. This law allows the EMA to assess the quality, safety and efficacy of a medicine or vaccine and its risk-benefit balance even though it will not be marketed in the EU. Instead, the EMA helps to facilitate access to new medicines for people living outside of the EU.
In October 2015 two World Health Organization (WHO) advisory groups – the Strategic Advisory Group of Experts (SAGE) on Immunization and the Malaria Policy Advisory Group (MPAG) – jointly called for pilot implementation of the vaccine in three to five settings in sub-Saharan Africa. The WHO officially adopted this position in January 2016.
2018: RNA proves promising
A team of researchers at Yale University collaborated with Novartis to test out an RNA vaccine against malaria. They took a strain of the malaria parasite with Plasmodium macrophage migration inhibitory factor (PMIF) genetically deleted, as PMIF suppresses T cells, a key part of the body’s immune response. They observed that mice infected with that strain developed memory T cells and showed stronger anti-parasite immunity.
The research team then used two mouse models of malaria to test the effectiveness of a vaccine using PMIF. One model had an early-stage liver infection from the Plasmodium, and the other a severe, late-stage blood infection. In both models, the vaccine protected against reinfection.
As a final test, the researchers transferred memory T cells from the immunized mice to “naïve” mice never exposed to malaria. Those mice were also protected.
2019: RTS,S rollout begins
Beginning in 2019, three sub-Saharan African countries – Ghana, Kenya and Malawi – have led the introduction of the vaccine in selected areas of moderate-to-high malaria transmission as part of a WHO-led pilot programme.
While the efficacy of RTS,S was established during the Phase III trial, the Malaria Vaccine Implementation Programme (MVIP) will assess the feasibility of administering the recommended four doses.
It will also assess the vaccine’s potential role in reducing childhood deaths and its safety during routine use. This will be used to inform the WHO’s policy on broader recommendation of the vaccine.
January 2021: GSK, MIV and Bahrat Biotech agree to supply RTS,S long-term
In January this year, GSK, MIV and Bakrat Biotech signed a product transfer agreement to help ensure the long-term supply of RTS,S. The manufacturing of the antigen part of the RTS,S vaccine will be transferred to Bahrat Biotech, alongside the grant of a license on all rights pertaining to the vaccine. GSK will retain the production of the adjuvant of the vaccine (AS01E) and will supply it to BBIL.
Bahrat Biotech chairman and managing director Krishna Ella said: “We salute the commitment by GSK, PATH, and partners, in developing a novel vaccine against malaria, a dreaded disease with more than 200 million cases worldwide.
“We are truly honoured to support this partnership and to provide global access to RTS,S/AS01. With prior expertise in malaria research, WHO prequalified vaccines, supplied to more than 70 countries, Bharat Biotech is geared up for large-scale manufacturing, and to provide continuous long-term supply of this life-saving vaccine.”
February 2021: Yale researchers patent RNA approach
After applying for a vaccine patent for the novel RNA vaccine to protect against the malaria parasite, the Yale researchers finally had the patent published by the US Patent & Trademark office.
April 2021: A challenger arises
Researchers from the University of Oxford reported the findings of a Phase IIb trial of a new potential malaria vaccine, R21/Matrix-M, which demonstrated an efficacy of 77% over a 12-month follow-up period.
R21 has been trialled in 450 participants aged five to 17 months in Nanoro, Burkina Faso. The participants were split into three groups, with the first two groups receiving the R21/Matrix-M (with either a low dose or high dose of the Matrix-M adjuvant) and the third receiving a rabies vaccine as the control group. Doses were administered from early May 2019 to early August 2019, largely prior to the peak malaria season.
The researchers were able to report a vaccine efficacy of 77% in the higher-dose adjuvant group, and 71% in the lower-dose adjuvant group. No serious adverse effects related to the vaccine were noted. Researchers will continue to administer booster shots and follow the 450 participants for at least another year.
The researchers have now started recruitment for a Phase III trial to asses large scale safety and efficacy in 4,800 children aged five to 36 months across four African countries. While Burkina Faso’s malaria season lasts only around six months, the researchers now want to trial the drug in areas where malaria is a larger threat.
R21 is a modified form of RTS,S, which is designed to be both more potent and cheaper to produce. It uses the same CSP antigen, but at a higher proportion, and uses the Matrix-M adjuvant also utilised in the Novavax Covid-19 vaccine.
If confirmed in the Phase III trial, R21 could be the first malaria vaccine to bypass the WHO vaccine efficacy target of 75%. If the vaccine is approved, over 200 million doses can be manufactured annually by the Serum Institute of India.




