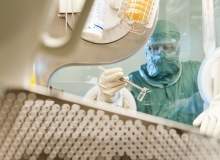
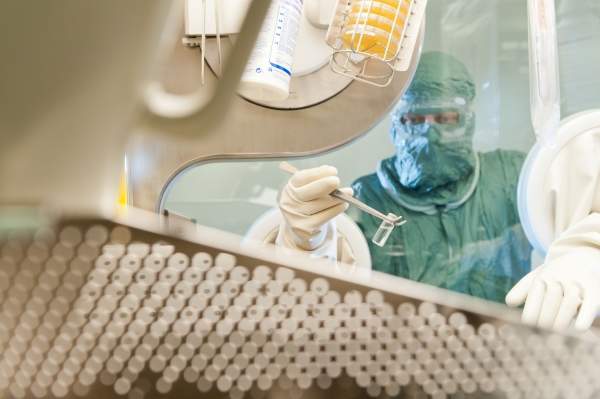
Roche’s new R&D facility is located in Wettsteinallee near Basel, Switzerland. It was officially inaugurated in November 2011. Roche made an investment of $271m in the Wettsteinallee site.
Designated as Building 97, the new facility replaces one of Roche’s older buildings – Building 60. The facility will be used for producing pharmaceutical formulations and also manufacturing samples for clinical trials.
The plant will employ 100 people who will be moved from Roche’s plants at other locations. It strengthens Roche’s presence in Basel where the company employs nearly 9,000 people.
Various functions of the pharmaceutical value chain already exist in Basel. The new plant will be an important addition to Roche’s facilities and establish Basel as a global research and development centre.
The new plant will also help in increasing Roche’s workforce in Basel. Several big pharmaceutical companies including Roche have been on a cost cutting spree due to increasing competition. Roche cut several sales and manufacturing jobs especially in the US in 2010.
Roche’s R&D facility details
Roche’s R&D activities are centred on developing a new medicine which is safe, tolerable and stable. Once a medicine is discovered, further research is carried out to market the drug in the form of a tablet, capsule, liquid or a pre-filled syringe.
Formulation is an important step in the development of a medicine as it enables it to be administered to patients. It also enables the active ingredient in the medicine to reach its target and optimises the amount of time it stays in the patient’s body.
The new facility will specialise in this research and help bring innovative drugs to the market. It will carry out R&D to transform new active substances into various solid and liquid dosage forms. The plant will also produce samples of new investigational drugs to be used in global clinical trials.
Specifications and design of Roche’s new research and development complex
The new R&D facility was designed to blend in with the existing buildings in Wettsteinallee. A glass facade similar to Roche’s other buildings was, therefore, not chosen for the plant.
The design of the facility is clear and compact featuring ribbon windows and white parapets. Its architecture is based on the Bauhaus tradition.
The northern part of the facility is spread over four storeys and is 20m high. The southern part includes nine storeys and is 40m high. Gross floor area of the building is 22,705m2.
The main structure of the R&D plant was constructed out of steel and reinforced concrete. It is capable of supporting heavy loads of up to 35t.
The plant includes 670 rooms including 239 clean rooms and features 42km of piping and 676km of cabling. It also includes three basement stories. The facility has been designed to be energy efficient and earthquake resistant.
Construction of Building 97 in Wettsteinallee, near Basel
Construction activities at the Wettsteinallee site commenced in August 2008 with the demolition of Building 60. In January 2009, Roche laid the foundation stone for the plant.
The plant required pouring of 32,500t of concrete, use of 1,750t of steel reinforcement and 530t of steel girders. For the construction of the basement, 25,000m3 of material was excavated. A major part of the supporting structure was prefabricated enabling construction to be completed quickly.
Contractors with a role in Roche’s Swiss R&D facility project
The main engineering contractor for the facility was M+W Process Industries. The company was also responsible for HVAC, process and mechanical engineering and clean room engineering.
Herzog & de Meuron was the architect for the project. The company also provided planning and construction development services.
Structural engineer for the facility was ZPF Ingenieure. Proplaning Architekten was the quantity surveyor.
Dr. Heinekamp Labor und Institutsplanung was responsible for laboratory engineering. IB Albrecht was responsible for plumbing engineering services. KIWI Systemingenieure & Berater, Dübendorf was responsible for providing electrical engineering services.
Emmer Pfenninger Partner was the facade consultant for the project and Zimmermann + Leuthe was responsible for the building physics.
Fire protection and safety consulting services were provided by Sicherheitsinstitut. Martin Lienhard was responsible for designing the building acoustics.