Siliconization is an important process in the manufacturing of many injectable drug products. These products are packaged in syringes, ampoules, auto-injectors or vials. Using a treatment to coat the internal surfaces of these items is a crucial step that manufacturers must take to improve the components’ safety, functionality, and processability.
To this end, the industry has been turning to a silicone-based fluid known as polydimethylsiloxane (PDMS) oil for decades. PDMS oils and emulsions act as lubricating agents for the internal surfaces of the packaging. While this may aid the efficiency of the manufacturing line (by facilitating the smooth insertion of a stopper into a vial, for example), the most important benefit of silicone’s hydrophobicity is the improved drainage of the container. Critically, this helps nurses and patients administer the correct dosage of medicine to patients.
Lubrication also ensures that all internal components in a prefilled syringe function as intended. The stopper of a syringe plunger could otherwise become ‘stuck’ to the glass surface of the barrel during storage, creating friction during the administration of the Active Pharmaceutical Ingredient (API). The result for the patient is an inefficient and potentially unpleasant experience. Siliconization reduces the forces required to administer the drug, improving the experience for the patient and caregiver.
There is also a vital safety reason that pharmaceutical companies deploy siliconization. This involves the need for a hydrophobic barrier between Active Pharmaceutical Ingredients (APIs) and glass packaging. Without such a barrier in place, interactions between the API itself and the packaging’s surface could occur, potentially altering the drug inside.
When serious safety risks are at play, it’s very important that manufacturers take siliconization seriously. This means not only choosing an appropriate silicone product, but also applying it correctly to each component. So, what are the requirements of a perfectly optimised layer of silicone fluid, and how can this be achieved?
Optimising siliconization
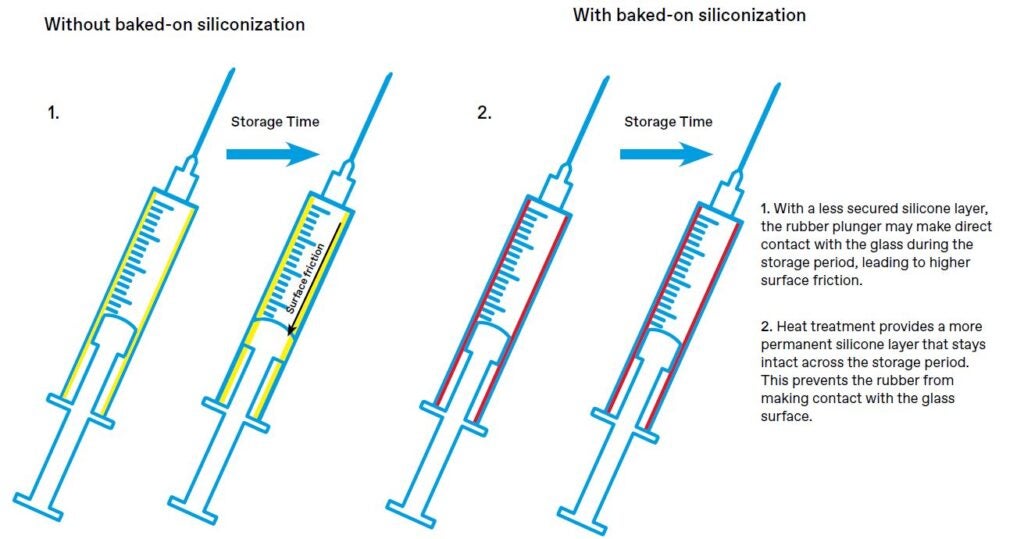
First things first, the layer must evenly coat all internal surfaces in contact with the drug product, leaving no areas uncovered. It must be robust and secure, enabling the silicone to stay in place for as long as necessary. Vials or syringes may be kept in storage for months at a time. If the internal components start to flow through the hydrophobic layer at any point, it could cause problems with functionality.
Finally, the quantity of silicone needs to be just right. Using too much could have adverse effects on the safety, quality, or purity of the drug product. When it comes to applying silicone fluids, glass and rubber components only require a thin film. Generally, a little goes a long way, hence why silicone-based emulsions can also be used in place of linear PDMS oils as an effective means of reducing the concentration level.
Applying the silicone layer
The methods used to apply the silicone coating typically include spraying, wiping or dipping. The coated article can then be air-dried or heated at an elevated temperature. The latter method refers to a process commonly known as “baked-on siliconization”. The benefit of “baking” the articles is the increased durability of the silicone layer, achieved through the removal of moisture and the possibility of oxidation and a degree of crosslinking between the silicone and the glass.
To eliminate any contamination risks, parenteral packaging components are always sterilized before the drug product is added. Ethylene oxide, steam autoclaving, dry heat sterilization, and low-dose gamma radiation have all been proven as acceptable techniques for sterilizing silicone-coated articles.
Quality control
There are a few analytical techniques available for quantifying the thickness of the silicone layer, although this can be challenging due to the low levels of fluid required (in a prefilled syringe barrel, this may be 0.2-1mg/barrel for sprayed-on silicone and 0.1mg/barrel for baked-on). [1]
Extraction of the silicone with organic solvents followed by FTIR spectroscopy is one method that can be applied after the spray-on siliconization step. This test can also be applied to devices with baked-on silicone layers. To determine the efficacy of the siliconization, users also perform functional tests such as contact angle or coefficient of friction measurements.
Although only a low level of silicone fluid is required, defining analytical methods for the quantification of the silicone layer is important for quality control. Stringent quality requirements govern the biopharmaceutical parenteral packaging industry. This also means drug developers should select high-quality fluids from trusted suppliers of pharmaceutical-grade silicones.
Developed by Elkem Silicones in accordance with the strict quality requirements of the pharmaceutical industry, Silbione™ FLD 70047 range (linear PDMS oils) and Silbione™ Emulsion (non-ionic emulsions of PDMS oil in water at 35%) are both great options.
Elkem Silicones is a leading global supplier of silicone materials for medical device and pharmaceutical applications. Many of the world’s leading pharmaceutical and biopharmaceutical companies trust Elkem to supply the silicone fluids they need to manufacture their injectable products safely and effectively.
To find out more about siliconization, download the whitepaper below.
[1] S. Funke. Optimization of the bake-on siliconization of cartridges. Part I: Optimization of the spray-on parameters.


