
Paradigm Health delivers a unified, AI-powered clinical research platform embedded within a national network of community and academic sites.
By grounding protocol design, patient identification, and study data capture in real-world patient population insights and care delivery, we enable faster enrollment, higher-quality data, and more efficient trial execution, making studies easier to run and expanding access to clinical trials for more patients, everywhere.
The Paradigm Health Clinical Trial Network
Paradigm Health provides access to a national network of community and academic research sites embedded in real-world care across the US, Japan, and Israel. An expansive footprint and deep site integration enables us to select the best sites for any trial.
Key stats:
- 800+ research sites
- 1,100+ specialists
- 2,100+ locations
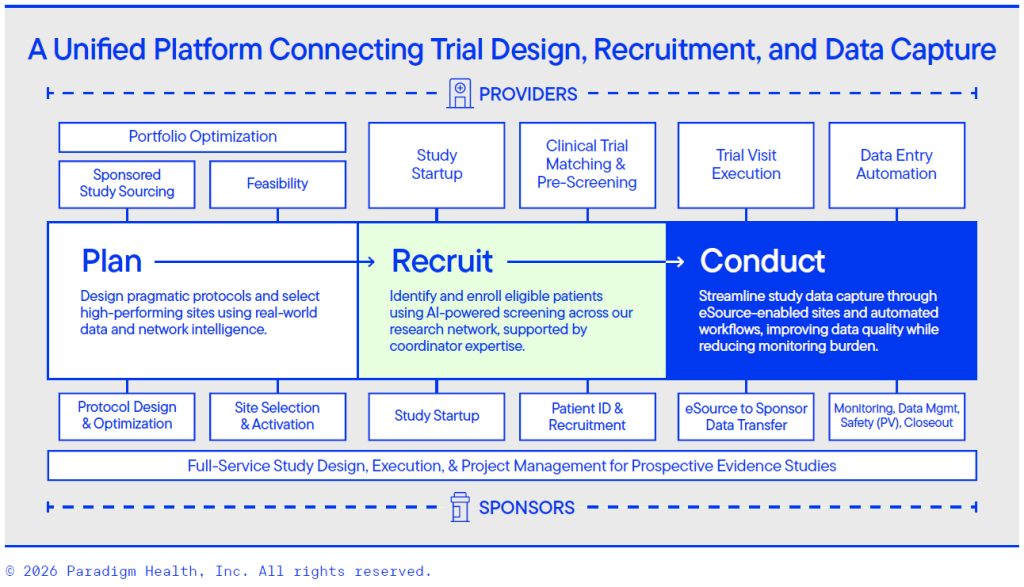
A platform delivering across the trial lifecycle
Plan
Trial design and protocol optimization: Model eligibility criteria against real-world clinical data to quantify impact on recruitable patient populations and identify design adjustments that improve feasibility and reduce costly amendments.
Feasibility and site selection: Evaluate sites using real patient availability, historical performance, and operational insights to identify those most likely to deliver sustained enrollment.
Recruit
Patient Identification and enrollment: Identify eligible patients directly within provider workflows using AI-powered screening across the network, supported by dedicated CRC expertise to confirm eligibility and guide enrollment.
Recruitment intelligence: Monitor recruitment performance across sites with visibility into screening volume, eligibility rates, and enrollment progress to identify bottlenecks and optimize site strategy.
Conduct
Automated source data capture: Capture clinical trial data directly from EHR systems and clinical documents to automatically populate eCRFs, reducing manual entry while improving data quality and speed.
Source data verification and monitoring: Enable efficient monitoring through source-linked data and automated traceability, reducing manual verification while lowering monitoring burden and cost.
Study conduct workflow management: Guide protocol execution through integrated workflows that streamline site operations, support investigator oversight, and reduce operational burden.
Embedded support for optimal trial performance
Paradigm Health’s AI-driven platform is paired with embedded support to provide human validation and additional site capacity.
- Drives patient progression from identification through enrollment
- Improves screening efficiency through clinical pre-review
- Ensures consistent follow-up regardless of site bandwidth
- Tracks and re-engages patients approaching eligibility
- Provides visibility into patient status and enrollment flow
Standards-based integrations across clinical and research systems
Paradigm Health connects clinical and research systems through a unified integration layer embedded within provider workflows, enabling scalable deployment across diverse environments.
- FHIR and HL7 based interoperability across EHR systems
- API-driven access to structured and unstructured clinical data
- Automated data mapping to EDC-aligned formats
- Direct EHR-to-EDC data flow with full source traceability
Scalable platform for integrated research and evidence (SPIRE)
Execute late-phase and post-approval studies directly within real-world care settings, combining embedded site workflows, continuous patient identification, and automated data capture.
To learn more about Paradigm Health’s solutions and how they are helping to address challenges with clinical research, download the document below.



