Two Ways to Minimize the Delamination Risk of Glass Containers
The recent spike in market recalls for injectable drug products due to visible flaky particles associated with 'glass delamination' demonstrates the need to solve these problems.

SCHOTT Pharma is a global leader in pharmaceutical packaging, focusing on primary drug containers and delivery systems for injectable medications.
You have successfully submitted your enquiry. Someone from our company will respond ASAP
Human health matters. That is why SCHOTT Pharma designs containment solutions grounded in science to ensure that medications are safe and easy to use for people around the world. Every minute, more than 30,000 people receive an injection packed in a SCHOTT Pharma product.
The portfolio comprises drug containment solutions and delivery systems for injectable drugs ranging from prefillable glass and polymer syringes to cartridges, vials, and ampoules. Every day, a team of around 4,800 people from over 65 nations works at SCHOTT Pharma to contribute to global health.
The company is represented in all main pharmaceutical hubs with 17 manufacturing sites in Europe, North and South America, and Asia.
With over 1,000 patents and technologies developed in-house and a state-of-the-art R&D centre in Switzerland, the company is focused on developing innovations for the future.
Currently, SCHOTT Pharma has over 1,800 customers, including the top 30 leading pharma manufacturers for injectable drugs and generated revenue of €986m in the financial year 2025. SCHOTT Pharma AG & Co. KGaA is headquartered in Mainz, Germany, and listed on the Frankfurt Stock Exchange as part of the SDAX. It is majority owned by SCHOTT AG, which is owned by the Carl Zeiss Foundation. At SCHOTT Pharma, we are committed to sustainable development for society and the environment.
Further information at www.schott-pharma.com.
SCHOTT Pharma’s core business is manufacturing advanced primary packaging for injectable drug products. The portfolio is divided into Drug Containment Solutions (e.g. vials, cartridges, ampoules) and Drug Delivery Systems (prefillable syringes and related devices).
All products are made to stringent quality standards using high-performance materials like Type I borosilicate glass and medical-grade cyclic olefin copolymer.
Prefillable syringes (PFS) serve as both the storage container and the delivery device for injectable drugs, supplied sterile and ready to fill. SCHOTT Pharma offers PFS in premium glass (the syriQ® line) as well as in advanced polymer (the SCHOTT TOPPAC® range). These syringes provide a highly stable, long-term drug storage solution and enable safe, convenient injections for patients and caregivers.
Whether made from glass or cyclic olefin copolymer (COC) polymer, the syringes are engineered to maintain drug stability (even for sensitive biologics or vaccines) and to withstand special conditions like deep-cold storage for mRNA-based therapies.
By eliminating most preparation steps (the syringe is pre-sterilised and ready to use), these systems help reduce the risk of dosing errors, contamination, and drug waste during administration.
Pharmaceutical cartridges are small cylindrical containers (for devices like pen injectors and pumps) that enable accurate multi-dose delivery. Crafted from high-quality glass tubing, SCHOTT Pharma’s cartridges are known for robust construction and exceptionally accurate dosing performance.
They fit a wide range of injection devices to ensure smooth operation and patient safety. SCHOTT Pharma provides ready-to-use cartridges under its cartriQ® brand, including specialised designs for different drug requirements. For instance, double-chamber cartridges allow convenient reconstitution of lyophilised drugs, and break-resistant cartridges withstand high pressure or shock during use.
Standard cartridges come in various sizes and can be delivered sterile to streamline filling operations. With precision manufacturing and strict tolerances, these cartridges deliver consistent doses and integrate seamlessly into devices like insulin pens or auto-injectors.
Glass vials are a cornerstone of SCHOTT Pharma’s product range, used to contain injectable liquids, lyophilised drugs, and vaccines. SCHOTT Pharma’s vials are made from Type I borosilicate glass, offering high chemical resistance to minimise drug-container interactions. This ensures that medications – including sensitive biologics – remain stable over shelf life without significant leachables or pH shifts.
The vials have tight dimensional tolerances and superior cosmetic quality to support efficient high-speed filling and secure container closure integrity. For example, the adaptiQ® system provides ready-to-use vials in nest-and-tub formats for streamlined fill-finish processing.
SCHOTT Pharma has also developed innovative vial solutions under its EVERIC® family to meet specialised needs. EVERIC® vials feature ultra-clean inner surfaces and other enhancements to improve drug stability and safety.
For instance, EVERIC® pure vials have a homogeneous inner glass surface for unmatched drug stability. There are also variants for specific applications under the EVERIC® concept – for example, vials for deep-frozen biologics, highly sensitive formulations, or unique identifiers to support track-and-trace of individual containers. This modular approach helps clients choose optimal vials for their needs while ensuring compatibility with standard filling lines.
SCHOTT Pharma produces classic glass ampoules, reflecting the company’s long heritage. Ampoules are sealed single-dose containers opened by breaking the neck. SCHOTT Pharma’s ampoules are made from Type I glass with high chemical resistance and precise dimensions, ensuring drug stability until the point of use. The company also offers safety-enhanced versions: easyOPC ampoules have a scored neck for easier, controlled opening, and Anti-Counterfeiting (AC) ampoules use unique ring markings to deter tampering and fraud.
Beyond its products, SCHOTT Pharma offers value-added services to support clients from early drug development through commercialisation. Experts assist with everything from packaging design and material selection to laboratory analytics and regulatory filings, ensuring the packaging works optimally with the drug and meets compliance requirements. The company also emphasises sustainability in packaging, collaborating with customers to reduce carbon footprint and adopt eco-friendly practices.
SCHOTT Pharma addresses emerging industry challenges through targeted packaging solutions aligned with current trends. For example, as biologic drugs and biosimilars become more common, the company drives container innovation to maintain their stability (minimising glass delamination, protein interactions, etc.).
It has also developed solutions for mRNA vaccines and other therapies requiring low-temperature storage, ensuring containers withstand ultra-cold conditions and freeze–thaw cycles without integrity loss. To facilitate self-administration, SCHOTT Pharma provides options for delivering large-volume biologics – such as optimised polymer syringe systems – that accommodate high-viscosity drugs and enable high-speed, quality-consistent filling.
Through continuous innovation in these areas, SCHOTT Pharma serves as a strategic partner to drug makers, helping overcome packaging hurdles as new therapies come to market.
SCHOTT Pharma prides itself on quality and technical innovation. All products are manufactured under rigorous quality management, with state-of-the-art automation and inspection ensuring consistency.
For example, SCHOTT Pharma’s syringe lines undergo 100% camera inspection to achieve extremely low defect rates, supporting an efficient fill-and-finish process. Backed by a deep research and development (R&D) heritage, the company continually improves materials and designs – from ultra-pure vial coatings to digital traceability features. Today, SCHOTT Pharma remains at the forefront of primary packaging technology.

The recent spike in market recalls for injectable drug products due to visible flaky particles associated with 'glass delamination' demonstrates the need to solve these problems.

Protecting medication from undesired interactions with the surrounding environment and preserving their efficacy during shelf life is one of the most pressing challenges for the pharmaceutical industry.

Fractography (fracture analysis) is an underestimated technique which makes it possible to objectively determine the true root cause of glass breakage.

The new vial concept offers pharmaceutical companies a choice of features that can be flexibly combined, addressing individual needs and requirements in the context of the containers themselves and the efficient processing of such primary packaging. The three respective features are introduced to the market by EVERIC™.

A critical issue that may occur with IV drugs is microbial contamination.

SCHOTT Pharma’s new facility in Lukacshaza, Hungary, will increase the company’s capacity to deliver high-value solutions.

SCHOTT Pharma will build a $371m pre-fillable syringe manufacturing facility in Wilson, North Carolina, expanding to the US market.

SCHOTT opened the first GMP-compliant pharmaceutical packaging production facility in Russia in May 2011. The plant is located at Zavolzhe near Nizhny Novgorod city in Russia.

Integrating primary packaging and device expertise: a co-hosted event by SCHOTT Pharma and SHL Medical.
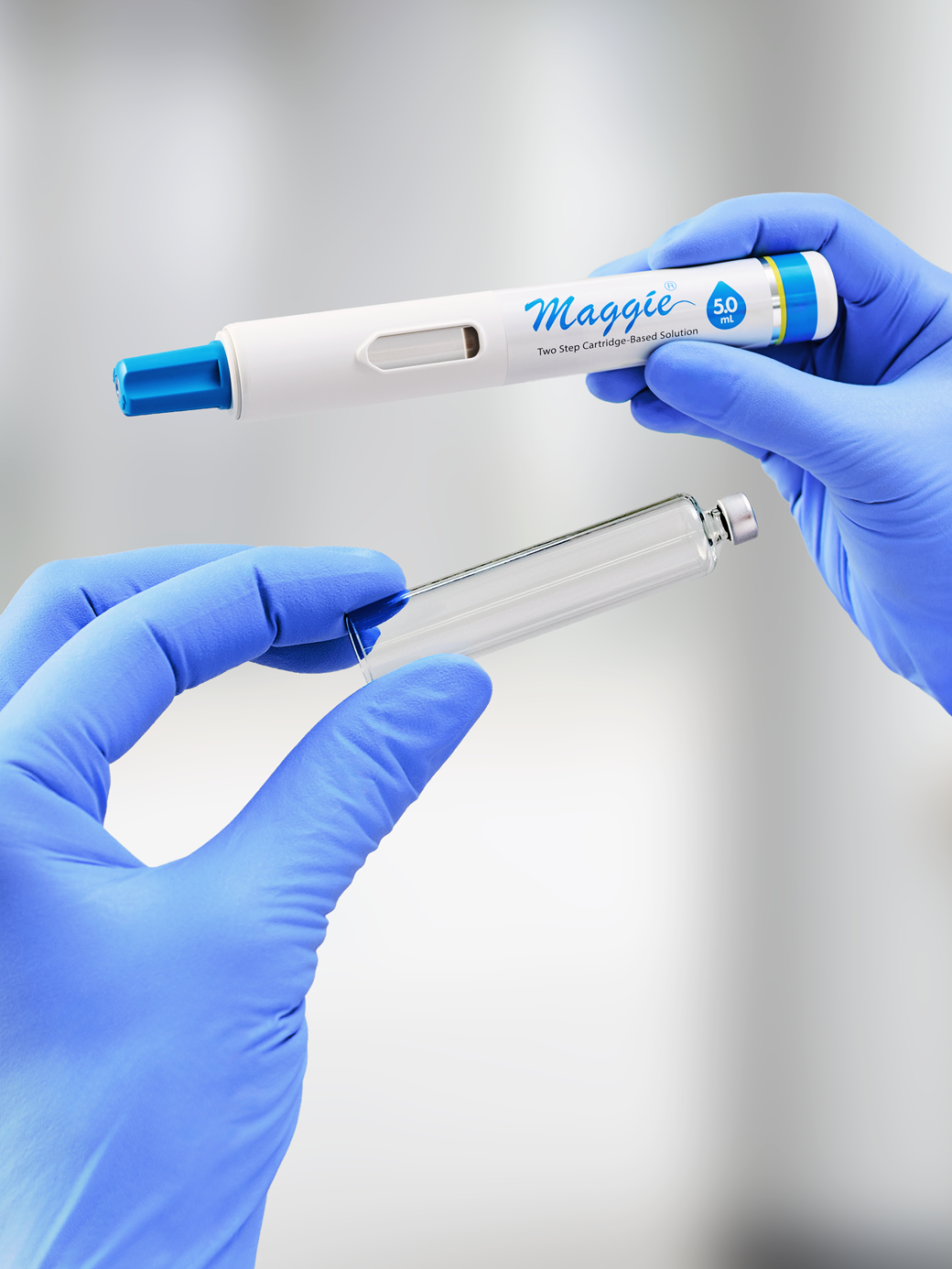
SCHOTT Pharma, a provider of drug containment and delivery solutions, is strengthening its collaboration with SHL Medical by completing compatibility pre-validation between the SCHOTT TOPPAC® polymer cartridge platform and SHL Medical’s Maggie® large-volume autoinjectors.

SCHOTT Pharma, a specialist in drug containment and delivery solutions, is expanding its EVERIC® portfolio with EVERIC® lyo & amber—an amber-colored vial made from FIOLAX® amber pharma glass and enhanced with the EVERIC® lyo hydrophobic inner coating.

Christian Mias, born in Iserlohn in 1974, will succeed Andreas Reisse starting May 1st.

The global large-volume injectors market is expected to witness substantial growth over the next decade, driven by factors such as the rising prevalence of chronic diseases, a shift towards homecare, and technological advancements in injector devices.

As the biologics market continues to expand, fuelled by breakthroughs such as cell and gene therapies, drug developers face increasing demand for safe, patient-friendly delivery and administration solutions.
As biologic therapies continue to rise for treating cancer, autoimmune, and neurological diseases, drug containment and delivery solutions are evolving to meet these new therapeutic demands.

The US Pharmacopeia (USP) has proposed a revision to General Chapter Container—Glass, which provides specifications for glass containers used in the pharmaceutical industry.

SCHOTT Pharma's prefilled syringes (PFS), whether made from glass or advanced polymer, provide a stable, long-term storage solution for drugs while offering a safe and convenient delivery system for both patients and clinicians. With fewer manual steps required compared to conventional packaging, PFS significantly lower the risk of medical errors and infections, prioritising the safety of healthcare professionals and patients alike.
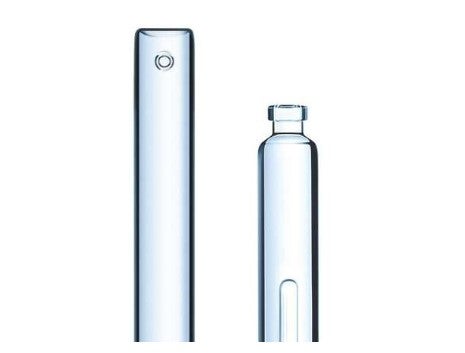
Robust, reliable, and compatible with a wide range of devices, SCHOTT Pharma's cartridge selection ensures precise drug dispensing across diverse medical scenarios.

Made of Type I Borosilicate Glass, SCHOTT Pharma's vials provide exceptional chemical resistance, safeguarding liquid drug formulations during storage. With precise dimensions and superior cosmetic quality, they facilitate an effective fill-and-finish procedure while ensuring container closure integrity.
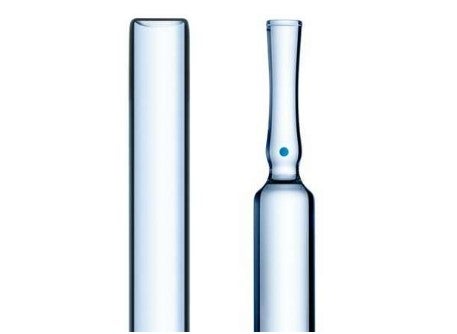
Manufactured using Type I Borosilicate Glass, SCHOTT Pharma ampoules offer high chemical resistance, accurate dimensions, tight cosmetic control, and drug stability from production to administration. With a focus on quality and safety, our ampoules provide the optimal solution for storing and delivering drugs with confidence.

SCHOTT Pharma offers tailored drug containment and delivery services, providing complete packaging solutions to meet your specific needs. Our comprehensive suite of services includes design, prototyping, and GMP-compliant documentation for primary and related packaging components.

SCHOTT Pharma offers data-driven analytical services that validate the suitability of containment solutions for your specific drug products. Our experts thoroughly assess material compliance, functionality, and drug-container interaction to ensure optimal container selection.

SCHOTT Pharma provides efficient fill-and-finish services, specialising in small-sample filling and closing during the early stages of production. Our expertise extends to conducting analytics to ensure seamless compatibility between packaging, containers, and line equipment.

SCHOTT Pharma provides comprehensive regulatory services to assist drug product and medical device owners in gathering the necessary information for registering containment systems. Our expertise ensures the global availability of information, complying with evolving international regulations, standards, as well as country-specific laws and guidelines.