Rethinking Pharma Packaging
Complex biological drugs offer hope for some of the hardest-to-treat conditions, from cancer to chronic disease. That promise comes with challenges. For example, biologics requires special packaging that will maintain the drug stability and ensure patient safety.
The structure of protein-based drugs is far more complex and variable than most generic drugs. This increases the risk of interaction with other materials, which can affect the active. On top of that, the drugs are highly viscous, making the administration more difficult and even causing discomfort or pain for the patient.
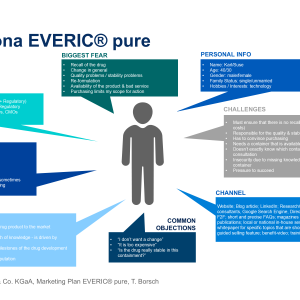
So what is essential for pharmaceutical containers that package highly sensitive drugs
for patients of all ages? Answers include:
- Improved drug stability
- Superior functionality
- Short time-to-market
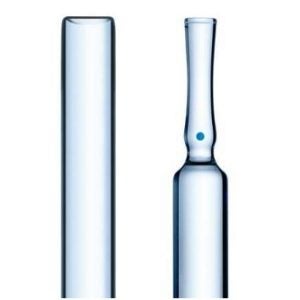
The solution: a new biotech syringe
SCHOTT recently developed syriQ BioPure® pre-fillable syringes that are designed for highly sensitive biopharmaceuticals. A uniform silicone layer, among others, ensures consistent gliding performance and full injection of the drug. The use of high-end materials further minimises the possible interaction between the drug and the container.
Accurate container dimensions are achieved through cutting edge converting technology based on accurate tubing. Each tube is closely inspected by SCHOTT’s big data perfeXion™ process, while the container is further examined throughout the forming process by online inspection systems. This enables seamless integration of syriQ BioPure® syringes in leading safety and autoinjector devices, allowing the patients to continue their treatment at home.
What’s next?
In the future, new drugs to treat diseases will be discovered. However, only if these are administered correctly can they help the patient. That is why researching syringes is an essential step in curing incurable diseases.