The pharmaceutical industry continues to be a hotbed of innovation, with activity driven by the evolution of new treatment paradigms, the gravity of unmet medical needs, as well as the growing importance of technologies such as pharmacogenomics, digital therapeutics, and artificial intelligence. In the last three years alone, there have been over 633,000 patents filed and granted in the pharmaceutical industry, according to GlobalData’s report on Innovation in Pharmaceuticals: Anti-bacterial compositions. Buy the report here.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
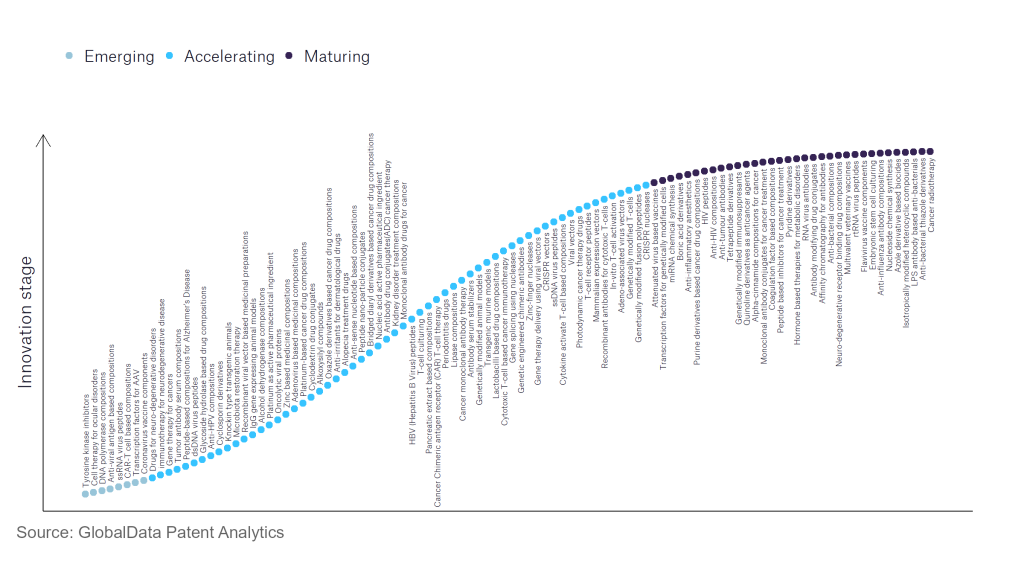
However, not all innovations are equal and nor do they follow a constant upward trend. Instead, their evolution takes the form of an S-shaped curve that reflects their typical lifecycle from early emergence to accelerating adoption, before finally stabilising and reaching maturity.
Identifying where a particular innovation is on this journey, especially those that are in the emerging and accelerating stages, is essential for understanding their current level of adoption and the likely future trajectory and impact they will have.
110 innovations will shape the pharmaceutical industry
According to GlobalData’s Technology Foresights, which plots the S-curve for the pharmaceutical industry using innovation intensity models built on over 756,000 patents, there are 110 innovation areas that will shape the future of the industry.
Within the emerging innovation stage, anti-viral antigen-based components, coronavirus vaccine components, and DNA polymerase compositions are disruptive technologies that are in the early stages of application and should be tracked closely. CRISPR nucleases, alcohol dehydrogenase compositions, and genetically modified fusion polypeptides are some of the accelerating innovation areas, where adoption has been steadily increasing. Among maturing innovation areas are anti-influenza antibody compositions and anti-bacterial compositions, which are now well established in the industry.
Innovation S-curve for the pharmaceutical industry
Anti-bacterial compositions is a key innovation area in pharmaceutical
Anti-bacterials are the subclass of antibiotics that kill or suppress the growth or reproducing ability of bacteria, which act by inhibiting cell wall synthesis, increasing cell membrane permeability, and interfering with their metabolism. Penicillin, cephalosporins, aminoglycosides, and macrolides are some of the examples of antibiotics/antibacterials.
GlobalData’s analysis also uncovers the companies at the forefront of each innovation area and assesses the potential reach and impact of their patenting activity across different applications and geographies. According to GlobalData, there are 140+ companies, spanning technology vendors, established pharmaceutical companies, and up-and-coming start-ups engaged in the development and application of anti-bacterial compositions.
Key players in anti-bacterial compositions – a disruptive innovation in the pharmaceutical industry
‘Application diversity’ measures the number of different applications identified for each relevant patent and broadly splits companies into either ‘niche’ or ‘diversified’ innovators.
‘Geographic reach’ refers to the number of different countries each relevant patent is registered in and reflects the breadth of geographic application intended, ranging from ‘global’ to ‘local’.
GSK is one of the leading patent filers in anti-bacterial compositions. It recently entered an exclusive licence agreement with Spero Therapeutics for the development of Spero’s late-stage antibiotic asset, tebipenem HBr. The company has reported positive data from a Phase II clinical trial of investigational antitubercular agent, GSK3036656, to treat patients with drug-susceptible pulmonary tuberculosis. According to the trial data, the drug GSK3036656 was found to be well tolerated and demonstrated EBA with a low, once-a-day oral dose.
To further understand the key themes and technologies disrupting the pharmaceutical industry, access GlobalData’s latest thematic research report on Pharmaceutical.
Data Insights
From

The gold standard of business intelligence.
Blending expert knowledge with cutting-edge technology, GlobalData’s unrivalled proprietary data will enable you to decode what’s happening in your market. You can make better informed decisions and gain a future-proof advantage over your competitors.



