The US Food and Drug Administration (FDA) has granted priority review for Danish biotechnology firm Genmab’s supplemental biologics licence application (sBLA) for ofatumumab (Arzerra), a human monoclonal antibody, to treat patients with relapsed chronic lymphocytic leukemia (CLL).
In Western countries, CLL is the most commonly diagnosed adult leukemia and accounts for about one in four case of the disease.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The sBLA was submitted to the FDA by Novartis under the company’s ofatumumab collaboration in July this year.
The FDA aims to complete its review of the ofatumumab sBLA within the time set by the Prescription Drug User Fee Act (PDUFA) and has been given a target date for completion of 21 January.
Genmab chief executive officer Jan van de Winkel said: "We are very pleased that the FDA has granted priority preview for ofatumumab, which means ofatumumab could potentially be available as a maintenance therapy for patients suffering from relapsed CLL relatively soon.
"We look forward to receiving FDA’s feedback on the application."
The sBLA is based on interim results from a Phase III trial, PROLONG (OMB112517), which evaluated ofatumumab maintenance therapy versus no further treatment in patients with a complete or partial response after second or third line treatment for CLL.
Ofatumumab is designed to target the CD20 molecule found on the surface of chronic lymphocytic leukemia (CLL) cells and normal B lymphocytes.
In the US, Arzerra is approved for use in combination with chlorambucil to treat previously untreated patients with CLL for whom fludarabine-based therapy is considered inappropriate.
The drug is approved in the EU for use in combination with chlorambucil or bendamustine for CLL patients who have not received prior therapy and are not eligible for fludarabine-based therapy.
Arzerra is also indicated in more than 50 countries as monotherapy to treat CLL patients who are refractory after prior treatment with fludarabine and alemtuzumab.
The company said that Arzerra is not approved anywhere as maintenance therapy for relapsed chronic lymphocytic leukemia.
Last November, Genmab entered an agreement for conditional transfer of the ofatumumab collaboration contract with GlaxoSmithKline (GSK) to Novartis.
The transfer of the collaboration follows an April 2014 GSK/Novartis Transaction, where Novartis agreed to acquire GSK’s oncology products including ofatumumab.
After the transfer, Novartis will develop and commercialise ofatumumab in oncology indications, while GSK will continue to develop and commercialise the drug for autoimmune indications.
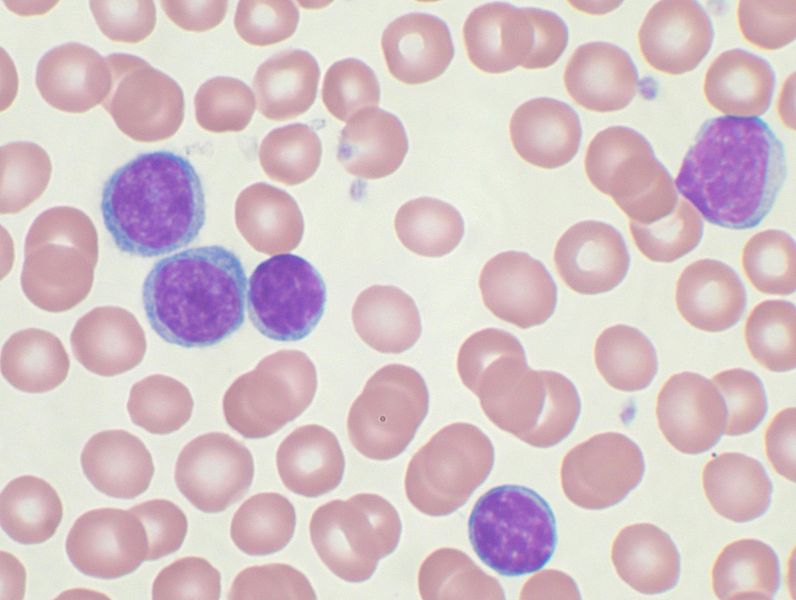
Image: Peripheral blood smear showing chronic lymphocytic leukemia cells. Photo: courtesy of Bibi Saint-Pol.




