Pacira BioSciences has entered a definitive agreement to acquire biopharmaceutical firm Flexion Therapeutics to boost its non-opioid pain portfolio.
The company will acquire Flexion for an equity value of nearly $450m or $630m, which includes debt.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Under the agreement signed by the companies, Flexion will receive $8.50 per share in cash along with one non-tradable contingent value right (CVR) worth up to $8.00 a share in cash if certain sales and regulatory milestones are met.
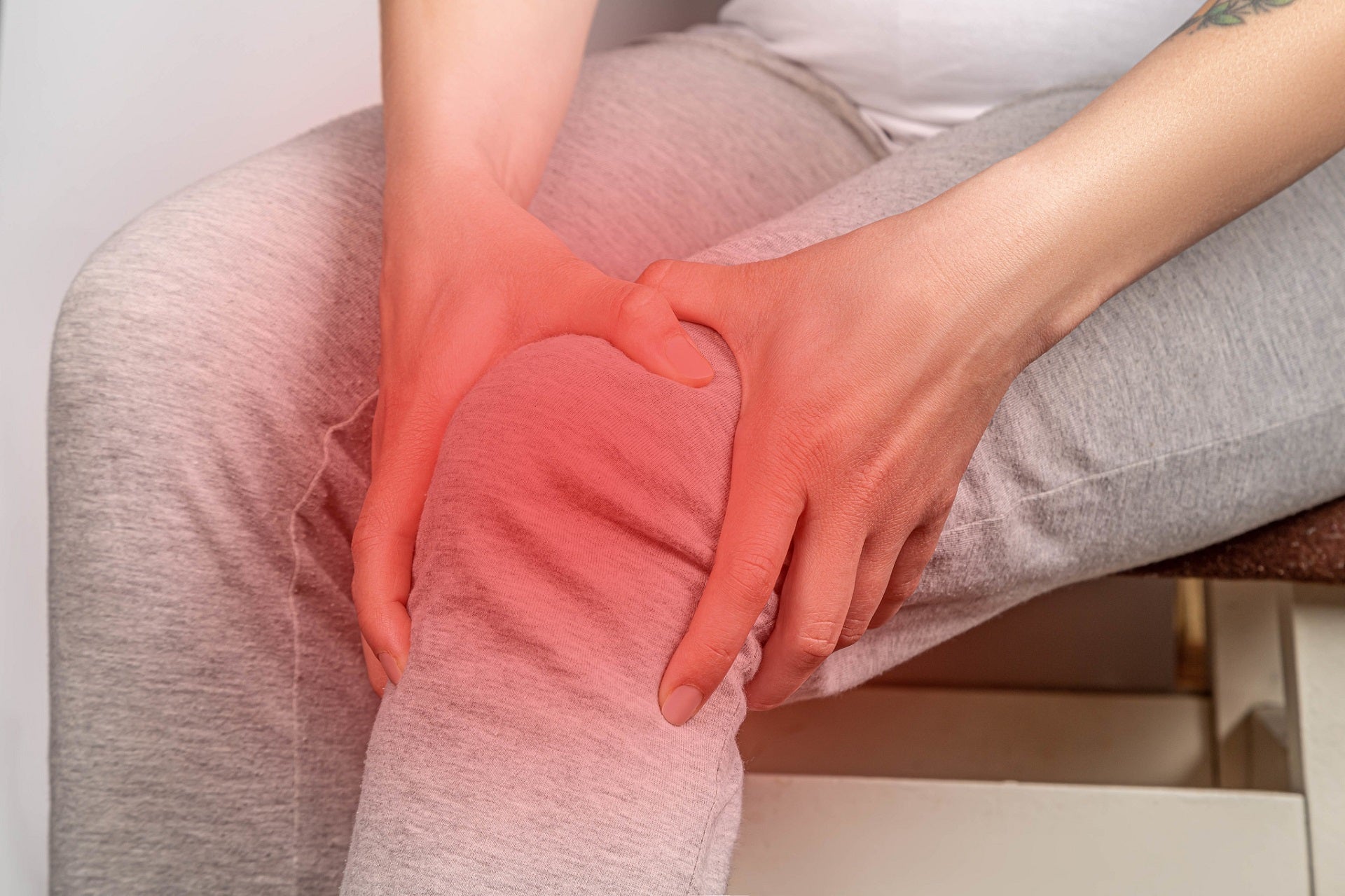
Through this acquisition, Pacira will add Flexion’s Zilretta, an FDA-approved treatment for osteoarthritis (OA) knee pain, to its commercial offering portfolio.
The therapy uses microsphere technology that combines triamcinolone acetonide with a poly lactic-co-glycolic acid (PLGA) matrix to provide extended pain relief.
Additionally, Pacira will get access to Flexion’s intra-articular gene therapy product candidate FX201 as well as FX301.
The transaction advances studies of FX201 to treat musculoskeletal pain and FX301 for postsurgical pain.
The combined portfolio will help Pacira in further expanding its position in non-opioid pain management. It will also help in providing end-to-end non-opioid solutions along the neural pain pathway.
Pacira BioSciences chairman and CEO Dave Stack said: “This acquisition is a major milestone in our strategy to build a robust offering of novel, non-opioid treatments to improve patient care along the neural pain pathway while simultaneously providing us with a complementary commercial asset in Zilretta for the treatment of OA knee pain.
“We believe the Flexion portfolio further solidifies Pacira as a leader in opioid-sparing pain management as we continue to redefine the role of opioids as a last resort rescue medication. Importantly, this acquisition creates diversification and growth to our topline while providing what we would expect to be meaningful synergies that should result in substantial near and long-term accretion to our cash flows and earnings.”
Subject to customary closing conditions, the transaction is anticipated to be completed in the fourth quarter of this year.



