
Syncona’s newly established portfolio company has signed a definitive agreement to acquire clinical-stage biotechnology company Freeline Therapeutics Holdings in an all-cash transaction.
The portfolio company will acquire all Freeline shares not currently owned by Syncona for $6.50 per American depositary share.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Freeline’s total issued share capital is therefore valued at $28.3m, a 51% premium compared to the closing price before Syncona’s initial proposal announcement on 18 October 2023.
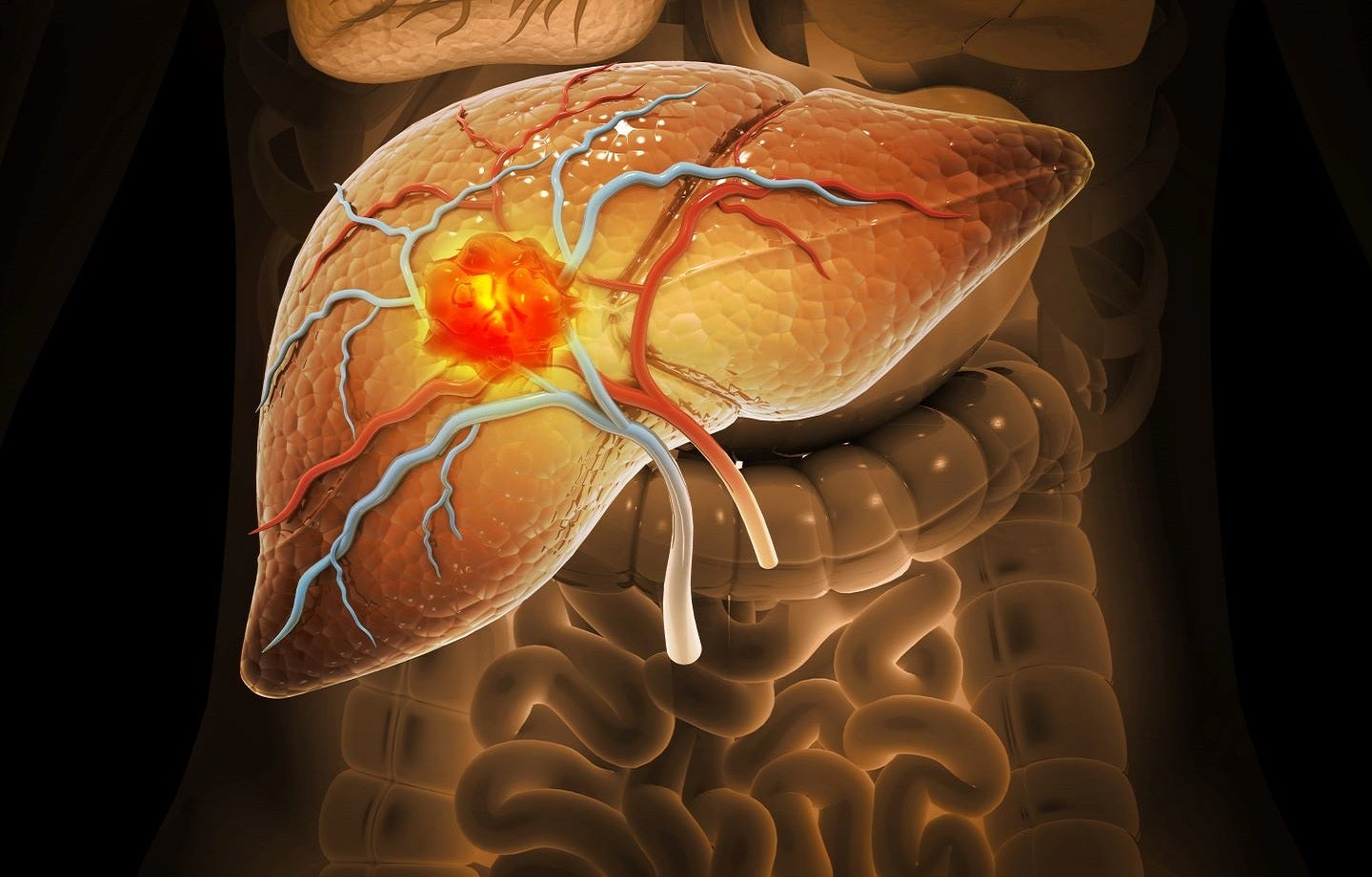
Freeline is focused on the development of innovative gene therapies for chronic debilitating diseases.
The company leverages its rationally designed AAV vector and capsid, AAVS3, and new promoters and transgenes to deliver a functional copy of a therapeutic gene into human liver cells.
It is currently advancing FLT201 gene therapy [a highly differentiated candidate] in a Phase I/II clinical trial in individuals with Gaucher disease type one. This therapy delivers a new transgene.
Freeline has other research programmes, including one focused on GBA1 [glycosylceramide beta 1]-linked Parkinson’s disease, which utilises the same new transgene as FLT201.
Freeline CEO Michael Parini stated: “Given the compelling data that we have seen from the first two patients treated with FLT201, we are committed to advancing FLT201 as expeditiously as possible, and we believe we will be better positioned to do that as a privately held, Syncona-backed company than we could by continuing as a publicly traded company in the current environment.”
Subject to the approval of Freeline’s minority shareholders, the deal is anticipated to be completed in the first quarter of 2024.
Syncona will offer Freeline secured convertible debt financing of up to $15m.
This funding will support operations and the ongoing development of FLT201 until the transaction is finalised.
Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva.
Editorial content is independently produced and follows the highest standards of journalistic integrity. Topic sponsors are not involved in the creation of editorial content.




