Amway, a direct-selling company based in the US, is constructing a new manufacturing facility at Quincy in Washington, US, for its NUTRILITE® Botanical Concentrates. The facility will process ingredients for nutritional products sold under its NUTRILITE brand of vitamins, minerals and dietary supplements.
The plant’s construction was started in August 2012 and is expected to be completed by the end of 2014. The total investment on the construction of the facility is estimated to be $38m. The plant is expected to create 30 direct / indirect jobs and about 100 construction jobs.
The new facility will process botanicals such as blueberry, Echinacea purpurea, Echinacea angustifolia, oregano, peppermint and nettle. The processed botanicals will be further supplied to Amway’s manufacturing facilities worldwide.
The Quincy facility is part of Amway’s $185m manufacturing expansion plan announced in 2012. The plan will help Amway to meet global demand for its NUTRILITE products.
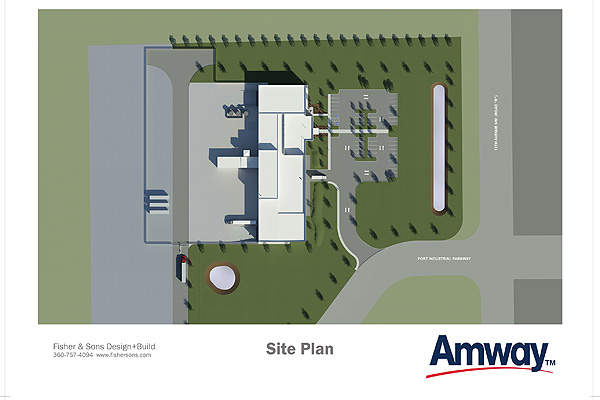
Details of Amway’s new manufacturing facility
Amway purchased 12 acres (4.85ha) of land from the Port of Quincy in April 2012. The Port of Quincy will allow the direct shipment of plant concentrates from the new facility to several nutritional product manufacturing facilities of Amway in California, China and India.
The new Amway NUTRILITE® facility will replace the current extraction and concentration operations in Lakeview, California. It will also support operations at Amway’s Trout Lake Farm, which is an organic herb farm located in Washington.
The plant will receive raw products from Amway’s Trout Lake Farms and process them into concentrates. The processed concentrates will further be transferred for vitamin manufacturing operations, globally.
The facility will cover an area of 48,000ft².
Processing at the botanical concentrate production facility
The new facility will produce concentrates which will be used in about two-thirds of NUTRILITE products. Extraction and concentration is an intermediary step in the process of transforming NUTRILITE seed into supplement.
Botanical concentrates will be extracted first, by pressing phytonutrient materials from freshly harvested or dehydrated crops to separate liquids from solids. The liquids will be evaporated from the extract to concentrate the phytonutrient solution. The concentrate solution will be then juiced or dried to form a powder.
NUTRILITE products made at Amway’s Quincy facility
Concentrates produced at the Quincy plant will be used in products such as NUTRILITE DOUBLE X® Vitamin/Mineral/phytonutrient, NUTRILITE Daily Multivitamin Multimineral and NUTRILITE Concentrated Fruits and Vegetables tablets.
Contractors involved with constructing the plant
Fisher & Sons is the design & build contractor for the project. University Mechanical will provide mechanical systems to process the raw materials at the facility. Schmitt Electric will serve as the electrical works contractor for the facility. Power Engineers will manage process controls at the plant.
Design and sustainability measures
The facility will be constructed of concrete panels. It will have a bar moist metal deck with a Thermoplastic Polyolefin (TPO) membrane roof. Fisher will install biochemical oxygen demand (BOD) monitors at the plant. The monitors will measure the BOD level in the wastewater before discharging it into the Port of Quincy after the wastewater treatment.
Marketing commentary for global pharmaceutical company Amway
Amway, which is headquartered at Ada in Michigan, US, has more than three million distributors spread across 100 countries across the world.
The company produces and markets consumer products through its agribusiness, manufacturing and logistics supply chain globally. It has 75 research and development laboratories spanning internationally.
The cooperative ranked as the world’s #1 direct selling business in the Direct Selling News Global 100, based on 2012 global sales of $11.3bn.
Nutritional product sales accounted for about 46% of Amway’s total sales in 2012. The top selling brands of the company include NUTRILITE, Artistry and eSpring.
Related content
Amway Nutrilite Vitamins Manufacturing Facility, Michigan, US
Amway, a direct-selling company based in the US, started the construction of a new Nutrilite® production facility in May 2013. The new facility is located at Spaulding Avenue, close to Ada in Michigan, US.
University of Washington (UW) Medicine Research Complex, Seattle, US
The new medical research complex at University of Washington (UW) School of Medicine in South Lake, Seattle was completed in March 2013.