Across the seven major markets, many drugs have been approved in Q1 2018. According to the GlobalData Drugs & Consensus Forecast database, Gilead Sciences’ Biktarvy (bictegravir + emtricitabine + tenofovir alafenamide) will be the top earner out of the products that received approval in 2018, with Johnson & Johnson’s Erleada (apalutamide) right behind it, through 2024.
Biktarvy is expected to make over $19B globally within the next seven years, and Erleada will make over $8B. This is due to the fact that human immunodeficiency virus (HIV) and prostate cancer treatments will be continuously needed through 2024.
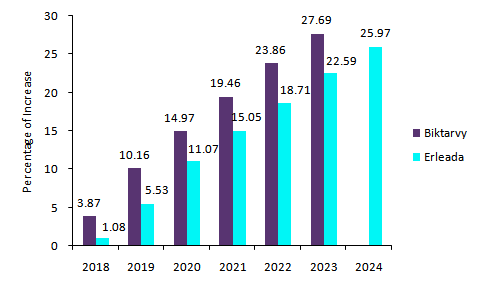
As shown in Figure 1 and Figure 2 below, there is an increase in percentage each year for both Biktarvy and Erleada through 2024, both globally and in the US, with Biktarvy expected to earn more each year.
Many drugs are expected to make over $1B from 2018–2014, including Vertex Pharmaceuticals’ Symdeko (tezacaftor + ivacaftor) and Kyowa Hakko Kirin’s Crysvita (burosumab). As it is only April, it is entirely possible that there could be further approvals of drugs that may make more revenue than Biktarvy and Erleada.
Two drugs that GlobalData expects to be approved in 2018 that may compete with Biktarvy and Erleada are Pfizer’s XTANDI (enzalutamide) for the treatment of prostate cancer and Merck’s doravirine for the treatment of HIV.

Figure 1: Global Drug Consensus Forecast 2018–2024. Credit: GlobalData.

Figure 2: US Drug Consensus Forecast 2018–2024. Credit: GlobalData.