Eligen® Technology Mechanism of Action
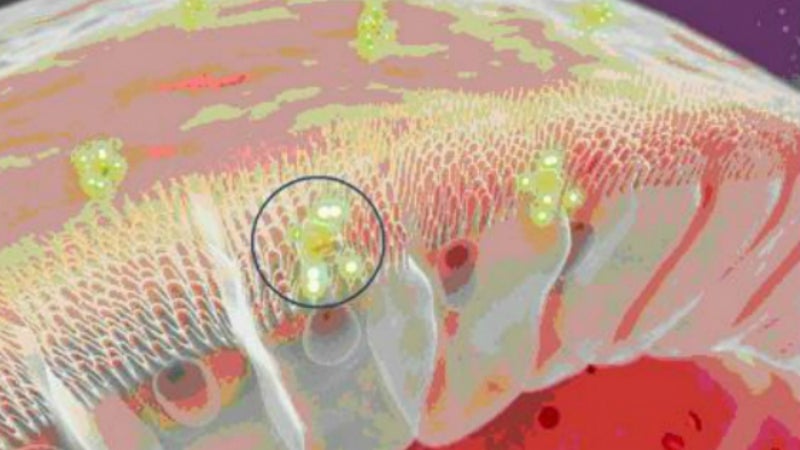
Emisphere's Eligen® Technology binds a delivery agent with a therapeutic compound without alternation. This process occurs when both agent and compound are in close proximity.

Emisphere Technologies is a drug delivery company that develops improved oral formulations of poorly permeable APIs such as peptides, proteins, oligonucleotides and polysaccharides, as well as polar small molecules.
You have successfully submitted your enquiry. Someone from our company will respond ASAP
Emisphere Technologies is a drug delivery company that develops improved oral formulations of poorly permeable active pharmaceutical ingredients (API) such as peptides, proteins, oligonucleotides, and polysaccharides, as well as polar small molecules.
Emisphere launched its first commercial product using their proprietary delivery system, Eligen Technology in March 2015.
Emisphere has developed a unique technology for enhancing the oral delivery of drugs that are normally given by injection or have poor oral bioavailability because their permeability through cell membranes is inadequate.
Eligen Technology has brought more than half a dozen oral formulations of peptides normally given by injection such as GLP-1 and insulin into clinical development.
In addition, oligosaccharides, polar small molecules and even metals have been shown to have significant increases in oral bioavailability in the clinic.
Eligen Technology uses small molecule carriers to orally deliver biologics and other normally impermeable drugs into the bloodstream.
These carriers help the drug molecules through the cell membranes of the epithelial cells lining the GI tract by a passive transport mechanism. Tight junctions remain intact so that no drug-drug interactions are observed, even when a second drug is given orally in a different dosage form at the same time.
The technology aims to alter the absorption phase, affecting the rate and extent, while the elimination phase is not affected.
In most cases, absorption is rapid with Tmax typically within an hour of dosing and can even occur within 30 minutes. Due to this attribute, Eligen can be used to create fast acting formulations.
Carrier and drug separate on reaching the bloodstream. The drug has been demonstrated to be in the same active form when injected, showing the expected pharmacological activity.
The carriers have an excellent safety profile. One of the carriers, SNAC, has been granted GRAS status. Other carriers in Emisphere’s library possess safety profiles which are amenable to becoming GRAS.
Because the carrier needs to be blended with the API, the manufacture of the dosage forms is simple. Eligen Technology employs only standard pharmaceutics processes, so scale-up is easy and inexpensive.
The technology works with both tablet and capsule solid dosage forms. Solutions and suspensions have also been demonstrated to be effective.
Emisphere’s drug delivery technology provides an improved convenience and compliance that an oral formulation brings.
In addition, oral dosing may improve safety and efficacy due to the ease of more frequent dosing compared to bolus dosing of biologics or small molecules.
The company can also bring new life to old products through lifecycle management using the registration of abbreviated procedures such as 505(b)2.
Many carriers in Emisphere’s library are NCEs and have a strong patent position. In addition, combination patents of drugs and carriers can provide further protection.
In a major milestone for oral drug delivery, Emisphere brought to market the first prescription product in a market that uses absorption enhancers of poorly permeable drugs.
In the first quarter of 2015, Emisphere launched Eligen B12 in the US. This product is directed to individuals with low serum B12 levels. In clinical testing Eligen B12 was shown to return B12 to normal levels in a manner that is comparable to a standard course of B12 injections.
Emisphere is partnered with Novo Nordisk in the area of diabetes. One of Novo Nordisk’s GLP-1 analogs is currently slated to begin Phase III development as an oral formulation using Emisphere’s drug delivery technology in 2016.
Novo Nordisk has recently expanded its relationship with Emisphere by licensing the technology for the oral delivery of four molecules targeting metabolic indications.
Emisphere also counts Novartis as a valued partner in which three peptides have been brought into development.
Please use the form below to send us your business enquiry.

Emisphere's Eligen® Technology binds a delivery agent with a therapeutic compound without alternation. This process occurs when both agent and compound are in close proximity.

Originally published by Elsevier Journals on 24 May 2011, this white paper outlines an open-label, randomised, single-dose, parallel-group study of the pharmacokinetics of oral cyanocobalamin formulated with Sodium N-[8-(2-hydroxy benzoyl) amino] caprylate (SNAC).

Novo Nordisk announced the European Commission (EC) has granted marketing authorisation for Rybelsus®, (oral semaglutide), for the treatment of adults with insufficiently controlled type 2 diabetes to improve glycemic control as an adjunct to diet and exercise.

Novo Nordisk announced the European Commission (EC) has granted marketing authorisation for Rybelsus®, (oral semaglutide), to treat adult patients with insufficiently controlled type 2 diabetes, improving glycemic control alongside diet and exercise.

Novo Nordisk has announced the Federal Food and Drug Administration (FDA) approved Rybelsus, a semaglutide tablet, to treat adults with type 2 diabetes.

Novo Nordisk has announced the submission of a marketing authorisation application (MAA) to the European Medicines Agency (EMA) for oral semaglutide, a glucagon-like peptide-1 (GLP-1) analogue tablet.

On 22-23 November, Novo Nordisk announced the completion and results of its Pioneer 9 and Pioneer 6 phase 3a trials, both conducted with an oral semaglutide, a once-daily oral formulation of the long-acting GLP-1.

Novo Nordisk has announced the results from Phase IIIa trial PIONEER 5, which investigated its oral semaglutide in adults with Type 2 diabetes and moderate renal impairment.

Novo Nordisk has announced it has completed the second Phase IIIa trial, PIONEER 2, with oral semaglutide, a once-daily oral formulation of the long-acting GLP-1 analog for the treatment of Type 2 diabetes, which utilises Emisphere Technologies' proprietary Eligen® Technology.

Emisphere Technologies has announced that it has reached an agreement with MHR Fund Management to waive certain terms of the company's existing obligations under the loan facility.

Emisphere Technologies has highlighted that Eligen licensee Novo Nordisk will initiate a global phase 3A development programme with oral semaglutide, a once daily oral formulation of the long-acting GLP-1 analogue semaglutide, for the treatment of type 2 diabetes.

Emisphere Technologies has announced it has entered a development and license agreement with Novo Nordisk to develop and commercialise oral formulations of four classes of Novo Nordisk's investigational molecules targeting major metabolic disorders, including diabetes and obesity, using Emisphere's oral Eligen technology.

Emisphere Technologies has announced US commercial availability of Eligen B12, the first and only once-daily oral prescription medical food tablet shown to normalise B12 levels without the need for an injection.

Emisphere Technologies has announced positive Phase II data from Eligen licensee Novo Nordisk concerning OG217SC.
Eligen B12™ is a once-daily oral non-prescription medical food shown to normalise B12 levels without the need for an injection.