While working in the pharmaceutical industry as a regional manager for the Caribbean and Central America, Dr Zulf Masters experienced first-hand the huge healthcare gaps present in emerging markets, especially regarding the availability of drugs.
This inspired Masters to set up his pharmaceutical company Masters Speciality Pharma in 1984, focusing on providing “emerging markets [with] access to trusted and quality medicines, as well as better treatments”. Ultimately he wanted to bridge the gap between the developed and developing world; the former has high quality medicines, and the latter has populations with widespread unmet needs.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Masters Speciality Pharma now supplies medicines to more than 90 countries worldwide, but currently focuses primarily on Latin America and the Middle East. It focuses on the fields of oncology, haematology, pain management and rare, orphan diseases.
Headquartered in the UK, the company has the regulatory oversight of relevant pharmaceutical bodies in the countries it works in, both in the developed and the developing world. “We have an understanding of local regulatory requirements,” says Masters. “Each of these countries has different requirements so our operation complexity are quite large, but we manage them successfully.
“The first thing we do when we enter a new country is see the regulator. We find out exactly what they want, what the regulations are, and how we can help.”
Masters Speciality Pharma has the support of a “network of distributor partners, regulatory consultants and lawyers for registration of products in each of the countries”.
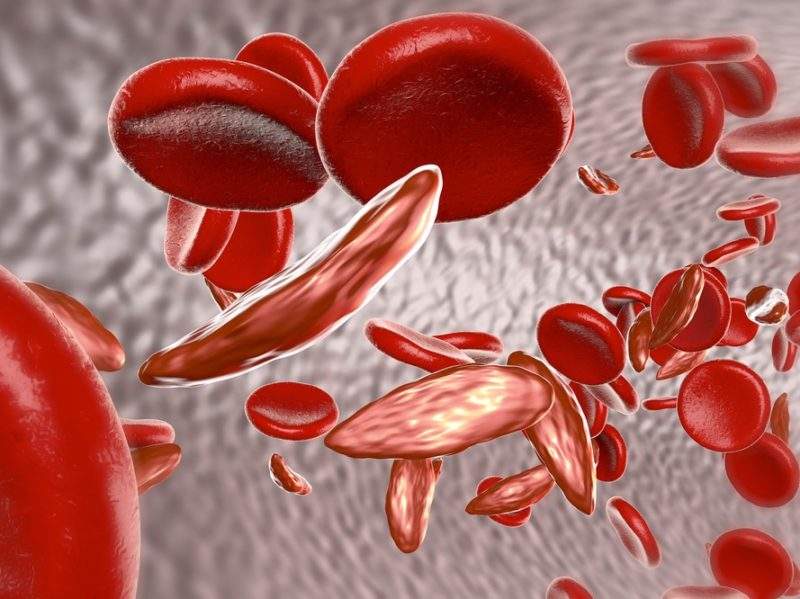
The company also vets and verifies all of its suppliers based on high standards of pharmacovigilance. One pharma company that has passed this screening process recently is French biopharma Addmedica. This led to Masters signing a deal to distribute Addmedica’s Siklos indicated for sickle cell disease, in Brazil.
Working with Addmedica to supply sickle cell drug in Brazil
Alongside other common conditions, such as Hepatitis C, Sickle cell disease is a major public health issue in Brazil. According to a 2011 report by Brazilian academics, 2,500 children are born every year with sickle cell disease in Brazil, with an especially high percentage of cases among the non-white populations living in the north-eastern regions and the states of São Paulo, Rio de Janeiro and Minas Gerais.
Masters comments: “We have been working in Brazil for over 20 years, and we were very aware of the fact that there are sickle cell disease patients in the North East of Brazil [who are] pretty untreated.”
Awareness of this large, unmet need contributed to Masters Speciality Pharma’s partnership agreement with Addmedica for Siklos. This will be the first drug used in Brazil to treat both children and adults with the disease.
Addmedica obtained European Commission marketing authorisation for the drug in 2017, making it the first pharma product indicated to prevent recurrent vaso-occlusive crises in anaemia associated with sickle cell disease. Siklos was recently launched in the US.
The success of Siklos to prevent these anaemia crises is because it can be offered in tailored dosing regimens. Its active pharmaceutical ingredient is hydroxycarbadmide, which blocks the growth and reproduction of cells, including blood cells, meaning it reduces the number of cells circulating the blood, as well as preventing some red blood cells from become sickled; its precise mechanism of action is unknown.
Masters notes: “So, Siklos, sickle cell disease and Brazil was a natural fit for us; to register the product in Brazil is an ethical and practical thing to do.” Masters Speciality Pharma was a good fit for Addmedica because, according to Masters, “they were looking for a trusted company that they could work with; because we are based in the UK, they had the confidence of working with us”.
Under the agreement, Masters Speciality Pharma will be responsible for registering the products with the Brazilian health authorities and will be the exclusive distributor in the country. They have collaborated with a “a very experienced regulatory lawyer in Brazil, [who] has already had meeting with Anvisa [Brazilian Health Regulatory Agency] and has continued to work with [the authorities] throughout the process”.
Masters says his company is looking to expand the Addmedica partnership to “other countries, such as Colombia, Costa Rica, Jamaica, and some countries in Africa” and potentially to other products Addmedica is developing, but this will be decided once the current projects reach completion.
Moving forward with Masters’ inclusive vision
In order to continue its efforts redressing the pharmaceutical industry’s neglect of the developing world despite the vast opportunities in the region, the company is seeking to develop ‘more partnerships in the territories where we work’.
Masters Speciality Pharma will also continue with the portion of its business that supplies drugs from larger countries within emerging regions to smaller ones; this is particularly common with drugs from larger South American countries being distributed to smaller Central American health systems.
An example noted by Masters was benznidazole, an antiparasitic medication for Latin American-specific Chagas disease, which is manufactured in Brazil. However, when Latin Americans emigrate, some carry the parasites in their blood and need urgent treatment once outside of the region. When this occurs Masters Speciality Pharma distributes benznidazole to the relevant country on the request of the World Health Organisation and the US Centres for Disease Control and Prevention.
Also, Masters notes the company plans to “increase our geographical reach” and move deeper into Latin America, as well as enter African markets.
However, Masters made it clear that in terms of conditions, Masters Speciality Pharma does not want to enter the primary healthcare market and will only work in “secondary healthcare for specialist products” in the areas of haematology, oncology, pain management and rare diseases.




