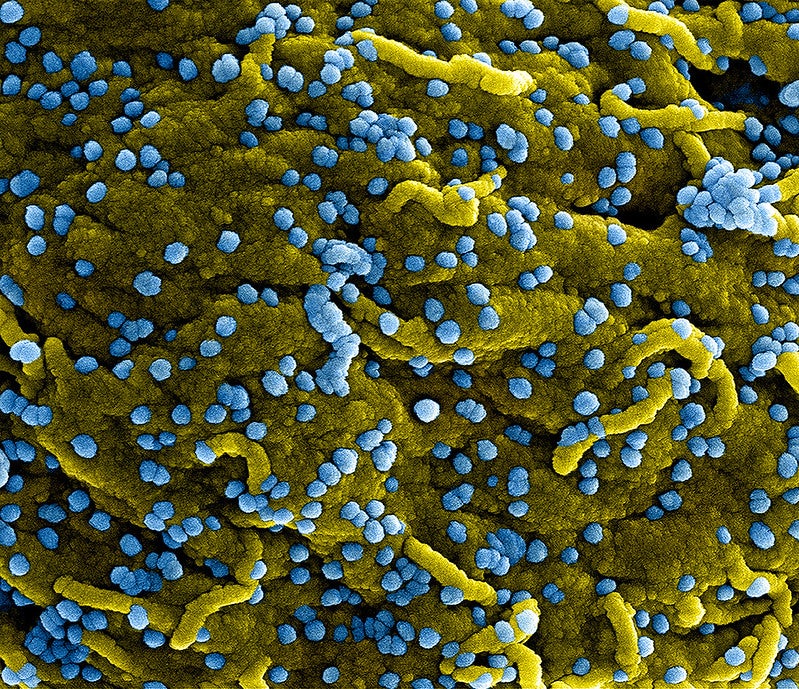
Amid the search for cure and treatments, China is testing the use of AbbVie’s HIV drugs to treat symptoms of the new coronavirus, which has killed 80 people to date.
According to the company, health authorities in China are using Kaletra drug, which contains a combination of lopinavir and ritonavir, for the treatment of infected patients.
The government notes that currently there is no effective anti-viral medication, recommending two lopinavir and ritonavir tablets and a dose of nebulised alpha-interpheron twice daily.
Medical journal Lancet said that a clinical trial of ritonavir and lopinavir is being conducted to treat new coronavirus cases, reported Bloomberg.
Sirnaomics is among the pharmaceutical companies that have commenced activities to develop drugs for the Wuhan coronavirus. Sirnaomics has engaged its research teams in the US and China to develop RNA interference (RNAi)-based prophylactics and therapeutics.
Previously, the company used small interfering RNA (siRNA) drugs to treat SARS coronavirus, H5N1 influenza and other respiratory viral infections.
To address the virus, the company discovered siRNAs that selectively act on the genes required for the viral infection and replication.
The company intends to partner with laboratories having cell culture models of the infection to develop several siRNA drug candidates.
Sirnaomics CEO and president Patrick Lu said: “Our team’s in-depth knowledge and experience using RNAi technology to fight respiratory viral infection in the previous SARS outbreak will add on to our antiviral weaponry to fight SARI.
“The unique siRNA drug design and chemical modification, respiratory specific siRNA delivery formulation, and the handheld nebulizer device are providing a clear pathway for developing novel siRNA therapeutics and prophylactics against 2019-nCoV infection.”
Scientists at the National Institutes of Health (NIH), the US have started working towards a vaccine for the coronavirus.
The US government announced a flight between Wuhan and San Francisco to evacuate all American citizens from the stricken city on 28 January.





