In December 2025, the FDA approved the first-ever GLP-1 pill for chronic weight management - Novo Nordisk’s oral formulation of Wegovy - marking a new phase in the brand’s growth strategy. The approval establishes a dual-format Wegovy portfolio that extends the brand beyond its once-weekly injectable and marks a strategic pivot toward oral delivery to sustain growth as injectable sales begin to plateau.
The oral formulation delivers once-daily semaglutide for the same chronic weight-management indication as the injectable, but with distinct administration requirements. Patients must take the pill on an empty stomach with a small volume of water and delay food and concomitant medications, in contrast to the once-weekly subcutaneous injection, which carries no such timing constraints. While the injectable retains convenience advantages in dosing frequency, the pill addresses needle aversion and may facilitate broader uptake in primary care settings, potentially expanding the overall treated population.
As illustrated in Figure 1, total Wegovy portfolio sales are forecast to increase from $13.5bn in 2026 to $18.9bn in 2031. Growth is strongest in the mid-forecast period before moderating toward 2031, indicating a shift from early launch acceleration to a more mature revenue trajectory.
Figure 1: Wegovy injection versus pill, total global sales forecast, 2026–2031
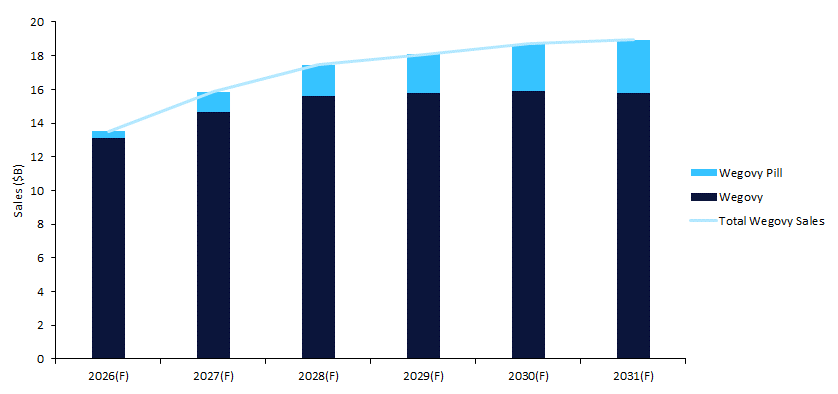
Injectable Wegovy sales are projected to rise from $13bn in 2026 to $15.9bn in 2030, before declining marginally to $15.8bn in 2031. Although the injectable remains the dominant revenue contributor throughout the forecast period, year-on-year expansion slows significantly after 2028, suggesting that the product is approaching a plateau phase. In the stacked columns shown in Figure 1, the injectable increasingly represents a large but stabilising revenue base for the brand.
By contrast, the Wegovy pill emerges as the key incremental growth driver. Between 2026 and 2031, total portfolio sales increase by $5.4bn (from $13.5bn to $18.9bn). Of this expansion, the oral formulation contributes $2.76bn - approximately 51% of incremental growth - while the injectable contributes $2.68bn, or 49%. Despite accounting for just 3% of total portfolio sales in 2026, the pill’s share is forecast to reach 17% by 2031. The chart, therefore, highlights a notable divergence: while the injectable maintains the larger absolute revenue base, the pill delivers a disproportionate share of forward growth.
These trends indicate that the future performance of the Wegovy franchise will increasingly depend on the uptake and positioning of the oral formulation, even as the injectable continues to anchor overall revenues. As injectable growth moderates and competitive pressures intensify in the obesity market, successful scaling of the pill will be critical in sustaining portfolio momentum and extending the brand’s lifecycle beyond its initial high-growth phase.