Softgel Capsule Production
Pharmagel is a leading reference point for worldwide softgel capsule production, thanks to its extraordinary growth in the pharmaceutical, nutraceutical and healthcare markets.

Pharmagel Technology develops Softgel Technology for pharmaceutical, nutraceutical and cosmetic products.
You have successfully submitted your enquiry. Someone from our company will respond ASAP

Pharmagel Technology develops Softgel Technology for pharmaceutical, nutraceutical and cosmetic products.
The softgel (soft gelatine) capsule is a one-piece, hermetically sealed shell wall filled with oils and non-aqueous liquids, plus solids in a paste or solution form.
The shell gives a unique strength and durability to softgels, and in addition protects the inner fill material from atmospheric oxidation that compromises other oral dosage forms in terms of potency and shelf-life.
Softgel has become widely used in prescription and OTC pharmaceuticals, plus also in nutraceuticals, because of the following advantages:
Pharmagel Technology is the leading supplier of semi-turnkey softgel encapsulation plants worldwide, having been engaged in the research and development of encapsulation lines and the formulation of softgels for four decades.
Pharmagel Technology’s softgel encapsulation equipment is used in 24 countries on 5 continents. Many of the plants have been completely redesigned and supplied with technology and training by Pharmagel Technology. This experience combined with our many decades of experience in the industrial production of softgels, enables Pharmagel Technology to give clients a unique and unrivalled guarantee of industrial manufacturing success in their own markets.
The Pharmagel Technology range of softgel encapsulation lines and ancillary equipment, combined with Pharmagel Technology’s technology and knowledge of the worldwide softgel market, offers clients a service of unrivalled quality and capability. That is especially important with the increasing use of softgels over the past two decades in the pharmaceutical, nutraceutical, cosmetic and leisure industries.
The MK3 range of Softgel lines represents a new generation of rotary die encapsulation machines designed to comply with international GMP and with the capability to make all capsule types, with a performance capability of up to 450 million softgels per year (typical 7.5 minimum oval size).
Fill and capsule base preparation – These two phases are the key for a successful encapsulation process. Using the example of the most complex fill type, complex pastes such as multivitamins /minerals or herbal extracts; these must be processed with proper specification mixing, refining and homogenising equipment and procedure, under vacuum. Particle size and viscosity are optimised for easy and accurate encapsulation. GMP equipment design with proven clean-in-place (CIP) systems is also vital to avoid cross-contamination between products.
Capsule base preparation is a critical process as it directly affects capsule shape, appearance and seam strength.
Encapsulation and drying – Encapsulation is obviously the most critical phase for the production of a perfect softgel capsule batch. The rotary die process, which has been perfected by Pharmagel Technology more than four decades, is a continuous single operation, because with the rotary die process the softgel capsule is formed and filled in a single step under conditions of low humidity and accurately controlled temperature.
Following encapsulation, softgels undergo drying – firstly fluid bed drying for approximately two hours followed by one to three days tray drying.
Inspection – With Pharmagel Technology, batches require little inspection and sorting with Pharmagel Technology’s automatic sorting machines specially designed to automatically inspect capsules at high speed and with minimal labour costs.
Pharmagel Technology guarantee – advanced technologies and equipment, GMP encapsulation plant design, accurate selection of capsule raw materials, intelligent choice of excipients, and experienced design of formulations.
We outline below our project technology transfer services:
Engineering
Technology

Pharmagel is a leading reference point for worldwide softgel capsule production, thanks to its extraordinary growth in the pharmaceutical, nutraceutical and healthcare markets.

Pharmagel's quality and reliability have been tested and accepted by some of the biggest pharmaceutical multinational companies, which have used Pharmagel's capsule production services and installed its technology lines.

In the last years, the market of the food and nutraceutical supplements has had a remarkable development in the sales. The research for new products and the optimization of the existing ones have pushed the producers to more and more complex and peculiar formulations. It was born the need to create

The productive efficiency of a Softgel Capsule Plant is based on a high number of aspects, and each of them is important and connected to the others.
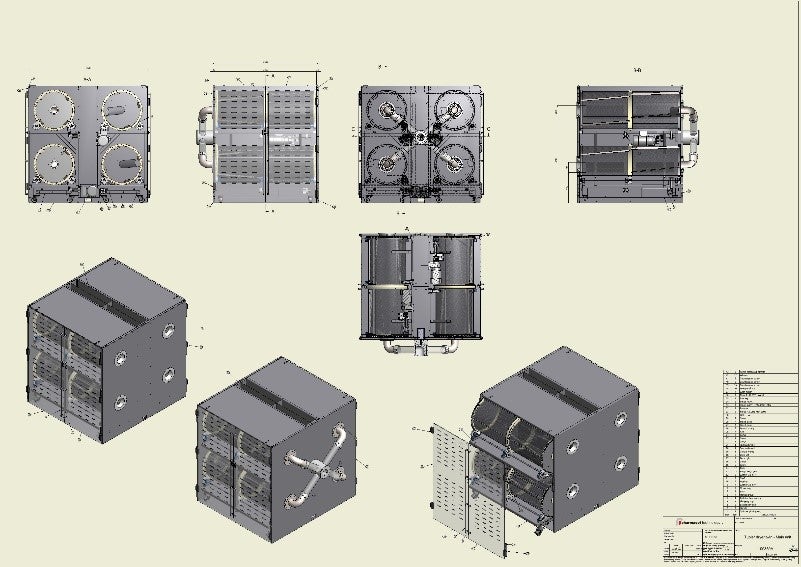
Pharmagel is a technological partner with global experience designing, building and commissioning standard and customised projects, with experience in softgel production combined with the supply of advanced equipment and technologies.

The pharmaceutical field that chooses to outsource their production to CMO is now equipping to produce industrial batches internally.
Pharmagel softgel capsule is the complete plant solution in Pharmaceuticals.